
SL Paper 2
Consider the following equilibrium.
\[\begin{array}{*{20}{l}} {{\text{2S}}{{\text{O}}_2}{\text{(g)}} + {{\text{O}}_2}{\text{(g)}} \rightleftharpoons {\text{2S}}{{\text{O}}_3}{\text{(g)}}}&{\Delta {H^\Theta } = - 198{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}} \end{array}\]
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for the reaction.
State and explain the effect of increasing the temperature on the yield of sulfur trioxide.
State the effect of a catalyst on the value of \({K_{\text{c}}}\).
State and explain the effect of a catalyst on the position of equilibrium.
Define oxidation in terms of oxidation numbers.
Describe using a labelled diagram, the essential components of an electrolytic cell.
Explain why solid sodium chloride does not conduct electricity but molten sodium chloride does.
Molten sodium chloride undergoes electrolysis in an electrolytic cell. For each electrode deduce the half-equation and state whether oxidation or reduction takes place. Deduce the equation of the overall cell reaction including state symbols.
Electrolysis has made it possible to obtain reactive metals such as aluminium from their ores, which has resulted in significant developments in engineering and technology. State one reason why aluminium is preferred to iron in many uses.
Outline two differences between an electrolytic cell and a voltaic cell.
Markscheme
\({\text{(}}{K_{\text{c}}} = {\text{)[S}}{{\text{O}}_{\text{3}}}{{\text{]}}^{\text{2}}}{\text{/[}}{{\text{O}}_{\text{2}}}{\text{][S}}{{\text{O}}_{\text{2}}}{{\text{]}}^{\text{2}}}\);
yield (of \({\text{S}}{{\text{O}}_{\text{3}}}\)) decreases;
forward reaction is exothermic / reverse/backwards reaction is endothermic / equilibrium shifts to absorb (some of) the heat;
Do not accept exothermic reaction or Le Châtelier’s Principle.
Do not allow ECF.
no effect;
no effect;
the rates of both the forward and reverse reactions increase equally;
increase in the oxidation number;
Annotated diagram of cell showing:
power supply/battery;
electrolyte;
cathode/negative electrode and anode/positive electrode;

(solid) ions in a lattice / ions cannot move;
(molten) ions mobile / ions free to move;
reduction occurs at the cathode/negative electrode and oxidation occurs at the anode/positive electrode;
Cathode/negative electrode: \({\text{N}}{{\text{a}}^ + } + {{\text{e}}^ - } \to {\text{Na}}\);
Anode/positive electrode: \({\text{2C}}{{\text{l}}^ - } \to {\text{C}}{{\text{l}}_2} + {\text{2}}{{\text{e}}^ - }/{\text{C}}{{\text{l}}^ - } \to \frac{1}{2}{\text{C}}{{\text{l}}_2} + {{\text{e}}^ - }\);
Award [1 max] if the two electrodes are not labelled/labelled incorrectly for the two half-equations.
Overall cell reaction: \({\text{N}}{{\text{a}}^ + }{\text{(1)}} + {\text{C}}{{\text{l}}^ - }{\text{(1)}} \to {\text{Na(1)}} + \frac{1}{2}{\text{C}}{{\text{l}}_2}{\text{(g)}}\)
Award [1] for correct equation and [1] for correct state symbols.
Allow NaCl(l) instead of Na+(l) and Cl–(l).
Al does not corrode/rust / Al is less dense/better conductor/more malleable;
Accept Al is a lighter (metal compared to Fe).
Accept converse argument.
electrolytic cell converts electrical energy to chemical energy and voltaic cell converts chemical energy to electrical energy / electrolytic cell uses electricity to carry out a (redox) chemical reaction and voltaic cell uses a (redox) chemical reaction to produce electricity / electrolytic cell requires a power supply and voltaic cell does not;
electrolytic cell involves a non-spontaneous (redox) reaction and voltaic cell involves a spontaneous (redox) reaction;
in an electrolytic cell, cathode is negative and anode is positive and vice-versa for a voltaic cell / electrolytic cell, anode is positive and voltaic cell, anode is negative / electrolytic cell, cathode is negative and voltaic cell, cathode is positive;
voltaic cell has two separate solutions and electrolytic cell has one solution / voltaic cell has salt bridge and electrolytic cell has no salt bridge;
electrolytic cell, oxidation occurs at the positive electrode/anode and voltaic cell, oxidation occurs at the negative electrode/anode and vice-versa;
Examiners report
Nearly all candidates deduced the equilibrium constant expression for the reaction given in (a) (i).
there were many good and complete answers here for (a) (ii). Some candidates did not state that the forward reaction was exothermic or the reverse reaction was endothermic, when trying to decide the effect of an increase in temperature on the yield of \({\text{S}}{{\text{O}}_{\text{3}}}\).
In (a) (iii) most candidates correctly stated that the catalyst would not have any effect on the value of \({K_{\text{c}}}\).
In part (iv) many candidates correctly stated that the catalyst would not have any effect on the position of equilibrium, but some did not explain why.
In (b) (i) some candidates defined oxidation as the loss of electrons but not in terms of oxidation numbers, as required by the question.
Some candidates described a voltaic cell instead of an electrolytic cell in (b) (ii). In some cases the electrodes were wrongly labelled or wrongly connected to the battery and the electrolyte was missing.
A large number of candidates stated that solid sodium chloride did not conduct electricity because it did not contain electrons in (iii). However some gave the correct answer indicating the free/moving ions as the particles responsible for the conductivity.
Part (b) (iv) was generally well answered. Most candidates lost a mark because they did not give the correct state symbols in the overall reaction.
Most candidates gave a correct answer as to why aluminium is preferred to iron in many uses in (b) (v).
There were very good answers indicating the main differences between an electrolytic cell and a voltaic cell in (vi).
To determine the enthalpy change of combustion of methanol, \({\text{C}}{{\text{H}}_{\text{3}}}{\text{OH}}\), 0.230 g of methanol was combusted in a spirit burner. The heat released increased the temperature of \({\text{50.0 c}}{{\text{m}}^{\text{3}}}\) of water from 24.5 °C to 45.8 °C.
The manufacture of gaseous methanol from CO and \({{\text{H}}_{\text{2}}}\) involves an equilibrium reaction.
\[{\text{CO(g)}} + {\text{2}}{{\text{H}}_2}{\text{(g)}} \rightleftharpoons {\text{C}}{{\text{H}}_3}{\text{OH(g)}}\,\,\,\,\,\Delta {H^\Theta } < 0\]
State and explain the effect of the following changes on the equilibrium position of the reaction in part (c).
Calculate the enthalpy change of combustion of methanol.
Using the theoretical value in Table 12 of the Data Booklet, discuss the experimental results, including one improvement that could be made.
Methanol can be produced according to the following equation.
\[{\text{CO(g)}} + {\text{2}}{{\text{H}}_2}{\text{(g)}} \to {\text{C}}{{\text{H}}_3}{\text{OH(l)}}\]
Calculate the standard enthalpy change of this reaction using the following data:
\[\begin{array}{*{20}{l}} {{\text{I: 2C}}{{\text{H}}_3}{\text{OH(l)}} + {\text{3}}{{\text{O}}_2}{\text{(g)}} \to {\text{2C}}{{\text{O}}_2}{\text{(g)}} + {\text{4}}{{\text{H}}_{\text{2}}}{\text{O(l)}}}&{\Delta {H^\Theta } = - 1452{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}} \\ {{\text{II: 2CO(g)}} + {{\text{O}}_2}{\text{(g)}} \to {\text{2C}}{{\text{O}}_2}{\text{(g)}}}&{\Delta {H^\Theta } = - 566{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}} \\ {{\text{III: 2}}{{\text{H}}_2}{\text{(g)}} + {{\text{O}}_2}{\text{(g)}} \to {\text{2}}{{\text{H}}_2}{\text{O(l)}}}&{\Delta {H^\Theta } = - 572{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}} \end{array}\]
Outline the characteristics of a chemical equilibrium.
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for this reaction.
Increase in temperature.
Increase in pressure.
Addition of a catalyst.
Markscheme
\((q = mc\Delta T = ){\text{ }}0.0500 \times 4.18 \times 21.3 = 4.45{\text{ (kJ)}}\);
Do not accept m = 0.05023 kg.
\({\text{(}}n{\text{ }}methanol = {\text{) }}\frac{{0.230}}{{{\text{32.05}}}} = 7.18 \times {10^{ - 3}}{\text{ (mol)}}\);
\(\Delta H = \frac{{4.45}}{{7.18 \times {{10}^{ - 3}}}}\);
\(\Delta H = - 6.20 \times {10^2}{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\);
Accept integer values of molar mass.
Final answer must have negative sign and correct units.
Award [4] for correct final answer with correct units.
less heat is liberated than theoretically/\( - 726{\text{ kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\);
probably due to heat loss/incomplete combustion;
determine heat capacity of calorimeter and take heat absorbed by calorimeter into account / any suitable insulation method / measure temperature with time and extrapolation of graph to compensate heat loss / OWTTE;
If the value calculated in (a) (i) is more exothermic than theoretically, allow ECF for M1 and for improvement if consistent.
\(\Delta {H^\Theta } = \frac{1}{2}{\text{II}} + {\text{III}} - \frac{1}{2}{\text{I}}\) / correct diagram/energy cycle;
\( - 283 - 572 - ( - 726)\);
\( - 129{\text{ (kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}{\text{)}}\);
Award [3] for correct final answer.
rate of forward reaction equals rate of backward reaction;
concentrations of reactants and products do not change / constant macroscopic properties;
\({K_{\text{c}}} = \frac{{{\text{[C}}{{\text{H}}_3}{\text{OH]}}}}{{{\text{[CO][}}{{\text{H}}_2}{{\text{]}}^2}}}\);
Do not award mark if incorrect brackets are used or brackets omitted.
shifts to left/reactants;
to endothermic side / (forward) reaction is exothermic;
shifts to the right/products;
to the side with fewer gas molecules/moles of gas;
no effect on equilibrium;
rate of forward and backward reaction increase equally / activation energy of forward and backward reaction lowered equally;
Examiners report
Many candidates used the mass of methanol in their calculation and most did not convert the mass of methanol to moles.
Students did not make a comparison between their calculated value and the theoretical value, often just stating the numbers. Most candidates were aware that heat was lost but improvements were generally simplistic.
The energy cycle was fairly well done, though working out could be shown better.
Many students had no problem with the characteristics of a chemical equilibrium.
The expression for\({K_{\text{c}}}\) was done quite well.
The effect of changes on the equilibrium position was answered quite well, though candidates had difficulty in explaining the rationale, omitting often gas molecules (ii) and increasing equally in (iii).
The effect of changes on the equilibrium position was answered quite well, though candidates had difficulty in explaining the rationale, omitting often gas molecules (ii) and increasing equally in (iii).
The effect of changes on the equilibrium position was answered quite well, though candidates had difficulty in explaining the rationale, omitting often gas molecules (ii) and increasing equally in (iii).
Biodiesel makes use of plants’ ability to fix atmospheric carbon by photosynthesis. Many companies and individuals are now using biodiesel as a fuel in order to reduce their carbon footprint. Biodiesel can be synthesized from vegetable oil according to the following reaction.

The reversible arrows in the equation indicate that the production of biodiesel is an equilibrium process.
Identify the organic functional group present in both vegetable oil and biodiesel.
For part of her extended essay investigation into the efficiency of the process, a student reacted a pure sample of a vegetable oil (where \({\text{R}} = {{\text{C}}_{{\text{17}}}}{{\text{H}}_{{\text{33}}}}\)) with methanol. The raw data recorded for the reaction is below.
\[\begin{array}{*{20}{l}} {{\text{Mass of oil}}}&{ = 1013.0{\text{ g}}} \\ {{\text{Mass of methanol}}}&{ = 200.0{\text{ g}}} \\ {{\text{Mass of sodium hydroxide}}}&{ = 3.5{\text{ g}}} \\ {{\text{Mass of biodiesel produced}}}&{ = 811.0{\text{ g}}} \end{array}\]
The relative molecular mass of the oil used by the student is 885.6. Calculate the amount (in moles) of the oil and the methanol used, and hence the amount (in moles) of excess methanol.
State what is meant by the term dynamic equilibrium.
Using the abbreviations [vegetable oil], [methanol], [glycerol] and [biodiesel] deduce the equilibrium constant expression \({\text{(}}{K_{\text{c}}}{\text{)}}\) for this reaction.
Suggest a reason why excess methanol is used in this process.
State and explain the effect that the addition of the sodium hydroxide catalyst will have on the position of equilibrium.
The reactants had to be stirred vigorously because they formed two distinct layers in the reaction vessel. Explain why they form two distinct layers and why stirring increases the rate of reaction.
Calculate the percentage yield of biodiesel obtained in this process.
Markscheme
ester;
amount of oil \( = \frac{{1013.0}}{{885.6}} = 1.144{\text{ mol}}\);
amount of methanol \( = \frac{{200.0}}{{32.05}} = 6.240{\text{ mol}}\);
since three mol of methanol react with one mol of vegetable oil the amount of excess methanol \( = 6.204 - (3 \times 1.144) = 2.808{\text{ mol}}\);
rate of the forward reaction is equal to the rate of the reverse reaction / forward and reverse reactions occur and the concentrations of the reactants and products do not change / OWTTE;
\({K_{\text{c}}} = \frac{{{\text{[glycerol]}} \times {{{\text{[biodiesel]}}}^3}}}{{{\text{[vegetable oil]}} \times {{{\text{[methanol]}}}^3}}}\);
to move the position of equilibrium to the right/product side / increase the yield of biodiesel;
no effect (on position of equilibrium);
increases the rate of the forward and the reverse reactions equally (so equilibrium reached quicker) / it lowers Ea for both the forward and reverse reactions by the same amount / OWTTE;
No ECF for explanation.
vegetable oil is mainly non-polar and methanol is polar / OWTTE;
stirring brings them into more contact with each other / increase the frequency of collisions / OWTTE;
Do not allow simply mixing.
(relative molecular mass of biodiesel, \({{\text{C}}_{19}}{{\text{H}}_{36}}{{\text{O}}_2} = 296.55\))
maximum yield of biodiesel \( = 3.432{\text{ mol}}/1018{\text{ g}}\);
percentage yield \(\frac{{811.0}}{{1018}} \times 100 = 79.67\% \);
Allow 80% for percentage yield.
Examiners report
Part (a) was reasonably well answered with most candidates opting for an ester. Ketone (frequently spelt keytone) and carbonyl were the most common incorrect responses.
In Part (b) most candidates scored 1 or 2 marks, showing that they knew the correct method but the third mark proved to be more difficult to obtain, usually because the factor of 3 was omitted.
In general, equilibrium (Part (c)) seems to be quite well understood. The most common error in (i) was to describe the reaction as constant rather than having opposing reactions with equal rates.
The expression in (ii) was an easy mark for the better candidates. The weaker ones often missed one or both of the powers of three and a small number had + signs in both the numerator and denominator.
In (iii) the most common incorrect answer was ‘to use up all the vegetable oil’.
In (iv) most candidates were aware that a catalyst has no effect on the equilibrium constant but failed to gain the second mark for saying that the catalyst affected both reactions equally, either by increasing the rates equally or lowering the activation energy by the same amount.
Very few candidates scored both marks for Part (d) of the question. The better candidates realised that there was a difference in polarity, though not always identifying which reactant was polar and which was non-polar. The most common answers either simply stated that the two were immiscible or that they had different densities. For the second mark an increase in collisions was often mentioned but not always an increase in the frequency of collisions.
Candidates found Part (e) to be very difficult. This was not helped by the small amount of space available to them on the paper. Many answers expressed the data in terms which would have calculated (100 - %) as though they had been drilled to calculate % impurities.
Methanol may be produced by the exothermic reaction of carbon monoxide gas and hydrogen gas.
\[\begin{array}{*{20}{l}} {{\text{CO(g)}} + {\text{2}}{{\text{H}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{C}}{{\text{H}}_{\text{3}}}{\text{OH(g)}}}&{\Delta {H^\Theta } = - 103{\text{ kJ}}} \end{array}\]
State and explain the effect of changing the following conditions on the amount of methanol present at equilibrium:
State the equilibrium constant expression, \({K_{\text{c}}}\), for the production of methanol.
increasing the temperature of the reaction at constant pressure.
increasing the pressure of the reaction at constant temperature.
The conditions used in industry during the production of methanol are a temperature of 450 °C and pressure of up to 220 atm. Explain why these conditions are used rather than those that could give an even greater amount of methanol.
A catalyst of copper mixed with zinc oxide and alumina is used in industry for this production of methanol. Explain the function of the catalyst.
Markscheme
\(({K_{\text{c}}}) = \frac{{{\text{[C}}{{\text{H}}_{\text{3}}}{\text{OH]}}}}{{{\text{[CO][}}{{\text{H}}_{\text{2}}}{{\text{]}}^{\text{2}}}}}\);
Do not award mark if incorrect brackets are used or brackets are missing.
amount (of methanol)/product decreases / less methanol;
(forward reaction) exothermic / reverse reaction endothermic / OWTTE;
amount (of methanol)/product increases / more methanol;
3 gas molecules/mol \( \to \) 1 / decrease in volume / fewer gas molecules on right
hand side/products / more gas molecules on left hand side/reactants;
high pressure expensive / greater cost of operating at high pressure;
lower temperature – lower (reaction) rate;
increases rate of forward and reverse reactions (equally) / lowers activation energy/\({E_{\text{a}}}\) (of both the forward and reverse reaction equally) / provides alternative path with lower activation energy/\({E_{\text{a}}}\);
Accept reactants adsorb onto the catalyst surface and bonds weaken resulting in a decrease in the activation energy.
Examiners report
Candidates were able to write an equilibrium expression accurately in part (a).
Candidates had a good understanding of the effect of temperature and pressure on an equilibrium system but some did not make reference to the change in the amount of methanol produced and hence did not always achieved full marks.
In (b) (ii) candidates also failed to achieve full marks if reference was not made to the gaseous nature of the reactants and products. Occasionally candidates incorrectly discussed the effect on the rate of reaction rather than answering the question.
There was much confusion with part (c) and candidates often struggled to express themselves adequately, demonstrating that they did not fully understand the question.
Part (d) on the other hand was extremely well answered with candidates having an excellent understanding of the function of a catalyst.
An example of a homogeneous reversible reaction is the reaction between hydrogen and iodine.
\[{{\text{H}}_{\text{2}}}{\text{(g)}} + {{\text{I}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{2HI(g)}}\]
Propane can be formed by the hydrogenation of propene.
\[{\text{C}}{{\text{H}}_3}{\text{CH=C}}{{\text{H}}_2}{\text{(g)}} + {{\text{H}}_2}{\text{(g)}} \to {\text{C}}{{\text{H}}_3}{\text{C}}{{\text{H}}_2}{\text{C}}{{\text{H}}_3}{\text{(g)}}\]
Outline the characteristics of a homogeneous chemical system that is in a state of equilibrium.
Deduce the expression for the equilibrium constant, \({K_{\text{c}}}\).
Predict what would happen to the position of equilibrium and the value of \({K_{\text{c}}}\) if the pressure is increased from 1 atm to 2 atm.
The value of \({K_{\text{c}}}\) at 500 K is 160 and the value of \({K_{\text{c}}}\) at 700 K is 54. Deduce what this information tells us about the enthalpy change of the forward reaction.
The reaction can be catalysed by adding platinum metal. State and explain what effect the addition of platinum would have on the value of the equilibrium constant.
State the conditions necessary for the hydrogenation reaction to occur.
Enthalpy changes can be determined using average bond enthalpies. Define the term average bond enthalpy.
Determine a value for the hydrogenation of propene using information from Table 10 of the Data Booklet.
Explain why the enthalpy of hydrogenation of propene is an exothermic process.
Describe a chemical test that could be used to distinguish between propane and propene. In each case state the result of the test.
Under certain conditions propene can polymerize to form poly(propene). State the type of polymerization taking place and draw a section of the polymer to represent the repeating unit.
Other than polymerization, state one reaction of alkenes which is of economic importance.
Markscheme
reactants and products in same phase/state;
rate of forward reaction = rate of reverse reaction;
concentrations of reactants and products remain constant / macroscopic properties remain constant;
Do not accept concentrations are equal.
\(({K_{\text{c}}}) = \frac{{{{{\text{[HI]}}}^{\text{2}}}}}{{{\text{[}}{{\text{H}}_{\text{2}}}{\text{][}}{{\text{I}}_{\text{2}}}{\text{]}}}}\);
no change to position of equilibrium;
no change to value of \({K_{\text{c}}}\);
the reaction is exothermic/heat is given out/ \(\Delta H\) is negative;
no effect (on the value of the equilibrium constant);
as it speeds up forward and reverse reaction / concentrations of reactants and products do not change / position of equilibrium does not change / no change in yield;
nickel / platinum / paladium;
150 − 200 °C/ heat;
Accept temperatures in this range.
Accept room temperature as an answer if platinum or palladium used.
the enthalpy change when (one mole of) the gaseous bond is broken (or formed) / \({\text{X}}–{\text{Y(g)}} \to {\text{X(g)}} + {\text{Y(g)}}/{\text{X(g)}} + {\text{Y(g)}} \to {\text{X}}–{\text{Y(g)}}\);
averaged for the same bond in a number of similar compounds / OWTTE;
energy in: C=C + H–H and energy out: C–C + 2C–H;
Accept energy in C–C + 6C–H + C=C + H–H and energy out 2C–C + 8C–H.
\(\Delta H = (612 + 436) - (347 + 826) = 1048 - 1173/ - 125{\text{ (kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}{\text{)}}\);
Award [2] for correct final answer.
Award [1] for +125.
If old Data Booklet values then allow: \(\Delta H = \) 1048−1172 = −124 (kJ\(\,\)mol–1)
due to the relative strength of the C–C and 2C–H bonds compared to the C=C and H–H bonds / bonds in products stronger than bonds in reactants;
(i) addition of bromine/bromine water;
the bromine colour remains with propane and propene decolourizes the bromine / solution changes from brown to colourless;
Do not accept “clear” instead of “colourless”.
addition (polymerization);
−(−CH(\({\text{C}}{{\text{H}}_{\text{3}}}\))−\({\text{C}}{{\text{H}}_{\text{2}}}\)−)− / −CH(\({\text{C}}{{\text{H}}_{\text{3}}}\))CH−;
Continuation bonds necessary for mark, displayed formula or condensed structural formula can be given.
Accept if more than one repeating unit is shown.
hydrogenation (of vegetable oils) / manufacture of margarine / manufacture of ethanol / addition of water;
Accept manufacture of alcohol.
Do not accept hydrogenation of alkenes.
Examiners report
Part (a) of this question focused on equilibrium and many candidates were able to show a good understanding of what would happen when the conditions were changed and were able to deduce the equilibrium expression. Most could describe the properties of a homogeneous equilibrium but some said that concentrations of reactants and products were equal at equilibrium as opposed to constant. The candidates also could state and explain the effect of a catalyst.
Part (a) of this question focused on equilibrium and many candidates were able to show a good understanding of what would happen when the conditions were changed and were able to deduce the equilibrium expression. Most could describe the properties of a homogeneous equilibrium but some said that concentrations of reactants and products were equal at equilibrium as opposed to constant. The candidates also could state and explain the effect of a catalyst.
Part (a) of this question focused on equilibrium and many candidates were able to show a good understanding of what would happen when the conditions were changed and were able to deduce the equilibrium expression. Most could describe the properties of a homogeneous equilibrium but some said that concentrations of reactants and products were equal at equilibrium as opposed to constant. The candidates also could state and explain the effect of a catalyst.
Part (a) of this question focused on equilibrium and many candidates were able to show a good understanding of what would happen when the conditions were changed and were able to deduce the equilibrium expression. Most could describe the properties of a homogeneous equilibrium but some said that concentrations of reactants and products were equal at equilibrium as opposed to constant. The candidates also could state and explain the effect of a catalyst.
Part (a) of this question focused on equilibrium and many candidates were able to show a good understanding of what would happen when the conditions were changed and were able to deduce the equilibrium expression. Most could describe the properties of a homogeneous equilibrium but some said that concentrations of reactants and products were equal at equilibrium as opposed to constant. The candidates also could state and explain the effect of a catalyst.
Part (b) proved more problematic and relatively few could describe the necessary conditions for hydrogenation, and even fewer could correctly state a definition of average bond enthalpy. The calculation of the bond enthalpy of propene proved difficult for many and although some gained marks by ecf few obtained the correct answer -125. Candidates also had difficulty explaining why the process was exothermic in terms of the relative strengths of the bonds being made and broken.
Part (b) proved more problematic and relatively few could describe the necessary conditions for hydrogenation, and even fewer could correctly state a definition of average bond enthalpy. The calculation of the bond enthalpy of propene proved difficult for many and although some gained marks by ecf few obtained the correct answer -125. Candidates also had difficulty explaining why the process was exothermic in terms of the relative strengths of the bonds being made and broken.
Part (b) proved more problematic and relatively few could describe the necessary conditions for hydrogenation, and even fewer could correctly state a definition of average bond enthalpy. The calculation of the bond enthalpy of propene proved difficult for many and although some gained marks by ecf few obtained the correct answer -125. Candidates also had difficulty explaining why the process was exothermic in terms of the relative strengths of the bonds being made and broken.
Part (b) proved more problematic and relatively few could describe the necessary conditions for hydrogenation, and even fewer could correctly state a definition of average bond enthalpy. The calculation of the bond enthalpy of propene proved difficult for many and although some gained marks by ecf few obtained the correct answer -125. Candidates also had difficulty explaining why the process was exothermic in terms of the relative strengths of the bonds being made and broken.
Part (c) was also based in organic chemistry and although most candidates could suggest bromine as a test for unsaturation, they did not all state a correct test result.
Candidates must make sure that they state that the bromine becomes colourless and not clear. Many realised that propene polymerises by addition polymerisation but few could successfully draw the structure of the repeating unit. Also few could suggest a reaction of alkenes of economic importance- such as hydration to make alcohols.
Part (c) was also based in organic chemistry and although most candidates could suggest bromine as a test for unsaturation, they did not all state a correct test result.
Candidates must make sure that they state that the bromine becomes colourless and not clear. Many realised that propene polymerises by addition polymerisation but few could successfully draw the structure of the repeating unit. Also few could suggest a reaction of alkenes of economic importance- such as hydration to make alcohols.
Part (c) was also based in organic chemistry and although most candidates could suggest bromine as a test for unsaturation, they did not all state a correct test result.
Candidates must make sure that they state that the bromine becomes colourless and not clear. Many realised that propene polymerises by addition polymerisation but few could successfully draw the structure of the repeating unit. Also few could suggest a reaction of alkenes of economic importance- such as hydration to make alcohols.
The Contact process involves an exothermic reversible reaction.
\({\text{2S}}{{\text{O}}_{\text{2}}}{\text{(g)}} + {{\text{O}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{2S}}{{\text{O}}_{\text{3}}}{\text{(g)}}\) \({K_{\text{c}}} \gg 1\) at 200 °C and 1 atm
Deduce the extent of the reaction at 200 °C and 1 atm.
The Contact process operates at a temperature of 450 °C and a pressure of 2 atm as optimum conditions for the production of \({\text{S}}{{\text{O}}_{\text{3}}}\). Outline the reasons for choosing these conditions.
Temperature:
Pressure:
An engineer at a Contact process plant hypothesized that using pure oxygen, instead of air, would increase the profits. Comment on whether or not her hypothesis is valid, giving your reasons.
Markscheme
concentration of products is much higher than the concentration of reactants / reaction nearly/almost goes to completion / position of equilibrium lies very far to the right / OWTTE;
Response must indicate the position of equilibrium is far to the right, but not complete conversion.
Temperature:
rate of reaction/production is slow at low temperature/fast at high temperature / OWTTE;
forward reaction is exothermic/backward reaction is endothermic
and
high temperature shifts equilibrium to left/reactants/favours reverse reaction / low temperature shifts equilibrium to right/products/favours forward reaction / OWTTE;
450 °C is a compromise temperature / produces a relatively good equilibrium yield at a reasonably fast rate;
Pressure:
rate of reaction/production is slow at low pressure/fast at high pressure / OWTTE;
more moles of gaseous reactants/less moles of gaseous products
and
high pressure shifts equilibrium to right/products/favours forward reaction / low pressure shifts equilibrium to left/reactants/favours reverse reaction / OWTTE;
a high yield/good reaction rate is obtained even at low pressure;
high pressure is expensive/dangerous;
(hypothesis is not valid as) equilibrium already nearly goes to completion / OWTTE;
(hypothesis is not valid as increase in yield may not be worth) expense of using pure oxygen / OWTTE;
(hypothesis is valid as pure oxygen) increases the rate of (the forward) reaction / more \({\text{S}}{{\text{O}}_{\text{3}}}\) produced per hour/day;
(hypothesis is valid as pure oxygen) shifts equilibrium to the right/products/\({\text{S}}{{\text{O}}_{\text{3}}}\) / increases the equilibrium concentration of \({\text{S}}{{\text{O}}_{\text{3}}}\);
Award [1 max] if no reference to “hypothesis”.
Examiners report
A number of candidates seemed confused as to what Part (a) required, but most students could relate completeness of reaction to the value of \({K_{\text{c}}}\), a common error being to declare the reaction “complete” rather than “almost complete”. Obviously some candidates had covered the “compromise” conditions for the reaction in some detail and could give a thorough answer to Part (b), though candidates often failed to give reasons (forward reaction exothermic and decreases moles of gas) for equilibrium shifts. Candidates seemed less at ease with the hypothesis question in Part (c), with many stating opinion without any reference to the hypothesis, in addition quite a few failed to realise that two separate factors were required to gain full marks.
A number of candidates seemed confused as to what Part (a) required, but most students could relate completeness of reaction to the value of \({K_{\text{c}}}\), a common error being to declare the reaction “complete” rather than “almost complete”. Obviously some candidates had covered the “compromise” conditions for the reaction in some detail and could give a thorough answer to Part (b), though candidates often failed to give reasons (forward reaction exothermic and decreases moles of gas) for equilibrium shifts. Candidates seemed less at ease with the hypothesis question in Part (c), with many stating opinion without any reference to the hypothesis, in addition quite a few failed to realise that two separate factors were required to gain full marks.
A number of candidates seemed confused as to what Part (a) required, but most students could relate completeness of reaction to the value of \({K_{\text{c}}}\), a common error being to declare the reaction “complete” rather than “almost complete”. Obviously some candidates had covered the “compromise” conditions for the reaction in some detail and could give a thorough answer to Part (b), though candidates often failed to give reasons (forward reaction exothermic and decreases moles of gas) for equilibrium shifts. Candidates seemed less at ease with the hypothesis question in Part (c), with many stating opinion without any reference to the hypothesis, in addition quite a few failed to realise that two separate factors were required to gain full marks.
When nitrogen gas and hydrogen gas are allowed to react in a closed container, the following equilibrium is established.
\[{{\text{N}}_{\text{2}}}{\text{(g)}} + {\text{3}}{{\text{H}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{2N}}{{\text{H}}_{\text{3}}}{\text{(g)}}\;\;\;\;\;\Delta H = - 92.6{\text{ kJ}}\]
Outline two characteristics of a reversible reaction in a state of dynamic equilibrium.
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for the reaction.
Predict, with a reason, how each of the following changes affects the position of equilibrium.
The volume of the container is increased.
Ammonia is removed from the equilibrium mixture.
Define the term activation energy, \({E_{\text{a}}}\).
Ammonia is manufactured by the Haber process in which iron is used as a catalyst. Explain the effect of a catalyst on the rate of reaction.
Sketch the Maxwell–Boltzmann energy distribution curve for a reaction, labelling both axes and showing the activation energy with and without a catalyst.
Typical conditions used in the Haber process are 500 °C and 200 atm, resulting in approximately 15% yield of ammonia.
(i) Explain why a temperature lower than 500 °C is not used.
(ii) Outline why a pressure higher than 200 atm is not often used.
Define the term base according to the Lewis theory.
Define the term weak base according to the Brønsted-Lowry theory.
Deduce the formulas of conjugate acid-base pairs in the reaction below.
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{N}}{{\text{H}}_{\text{2}}}{\text{(aq)}} + {{\text{H}}_{\text{2}}}{\text{O(l)}} \rightleftharpoons {\text{C}}{{\text{H}}_{\text{3}}}{\text{NH}}_{\text{3}}^ + {\text{(aq)}} + {\text{O}}{{\text{H}}^ - }{\text{(aq)}}\]

Outline an experiment and its results which could be used to distinguish between a strong base and a weak base.
Markscheme
rates of forward and reverse reactions are equal / opposing changes occur at equal rates;
the concentrations of all reactants and products remain constant / macroscopic properties remain constant;
closed/isolated system;
Accept “the same” for “equal” in M1 and for “constant” in M2.
\(({K_{\text{c}}} = )\frac{{{{{\text{[N}}{{\text{H}}_3}{\text{(g)]}}}^2}}}{{{\text{[}}{{\text{N}}_2}{\text{(g)]}} \times {{{\text{[}}{{\text{H}}_2}{\text{(g)]}}}^3}}}\);
Ignore state symbols.
Concentration must be represented by square brackets.
The volume of the container is increased:
position of equilibrium shifts to the left/reactants and fewer moles of gas on the right hand side/pressure decreases / OWTTE;
Ammonia is removed from the equilibrium mixture:
position of equilibrium shifts to the right/products and [NH3] decreases so [N2] and [H2] must also decrease to keep Kc constant
OR
position of equilibrium shifts to the right/products and rate of reverse reaction decreases / OWTTE;
Award [1 max] if both predicted changes are correct.
Do not accept “to increase [NH3]” or reference to LCP without explanation.
minimum energy needed (by reactants/colliding particles) to react/start/initiate a reaction;
Accept “energy difference between reactants and transition state”.
rate increases;
more effective/successful collisions per unit time / greater proportion of collisions effective;
alternative pathway and a lower activation energy
OR
lowers activation energy so that more particles have enough energy to react;
Do not accept just “lowers/reduces the activation energy”.
Accept “provides a surface for reacting/reactants/reaction”.
Curve showing:

general shape of Maxwell-Boltzmann energy distribution curve and labelled y-axis: probability of particles / frequency and labelled x-axis: (kinetic)energy;
Curve must begin at zero and must not cut the x-axis on the RHS.
Accept number/fraction/proportion of particles for y-axis label, but do not accept amount or just particles.
correct position of \({E_{\text{a}}}\) catalysed and \({E_{\text{a}}}\) uncatalysed;
Shading shown in the diagram is not required for the marks.
(i) slower rate / OWTTE;
uneconomic / OWTTE;
(ii) high cost for building/maintaining plant / high energy cost of compressor /OWTTE;
Do not accept “high pressure is expensive” without justification.
Accept high pressure requires high energy.
electron pair donor;
Accept lone pair donor.
proton acceptor and partially/slightly ionized;
Accept “proton acceptor and partially/slightly dissociated”.

Award [1 max] for two correct acids OR two correct conjugate bases.
solutions of equal concentration;
pH measurement/UIP;
strong base has higher pH;
OR
solutions of equal concentration;
electrical conductivity measurement;
strong base has higher electrical conductivity;
OR
solutions of equal concentration;
temperature difference in neutralization reaction with a strong acid;
strong base has a greater temperature difference;
Accept reverse arguments for observations.
Examiners report
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
This was, by far and away, the most common choice for Section B.
The conditions for an equilibrium system were well known, and the \({K_{\text{c}}}\) expression was almost universally correctly given, the incidence of curved brackets was very low. With the description of the effect of changing conditions, the increase in volume change generally scored, but the answers for the removal of ammonia were far too general to be given credit. It is pleasing to note that most candidates are aware of the importance of using the word “'minimum”, as well as the effect of a catalyst, with most giving perfect answers. The drawing of the Maxwell-Boltzmann energy distribution curve suffered from poor draughtsmanship. Too many curves did not start at the origin and lacked correct labels. An appreciable minority drew the energy/reaction co-ordinate graph. The knowledge of the compromise conditions for the Haber process was often confused, particularly with regard to why high pressure is not used, where far too many answers lacked the depth required. Occasionally the word “pair” was missing for the definition of a Lewis base, and with the definition of a weak Brønsted-Lowry base most candidates failed to appreciate the difference between partially/slightly ionized and “not completely” ionized, the part of proton acceptor was also often missed out. With the description of the experiment to show the difference between a strong and weak base, many scored two out of the three available; the concept of a fair test, and the importance of equal concentrations was rarely appreciated.
Group 7 of the periodic table contains a number of reactive elements such as chlorine, bromine and iodine.
Bleaches in which chlorine is the active ingredient are the most common, although some environmental groups have concerns about their use. In aqueous chlorine the equilibrium below produces chloric(I) acid (hypochlorous acid), HOCl, the active bleach.
\[{\text{C}}{{\text{l}}_2}{\text{(aq)}} + {{\text{H}}_2}{\text{O(l)}} \rightleftharpoons {\text{HOCl (aq)}} + {{\text{H}}^ + }{\text{(aq)}} + {\text{C}}{{\text{l}}^ - }{\text{(aq)}}\]
Aqueous sodium chlorate(I), NaOCl, the most common active ingredient in chlorine based bleaches, oxidizes coloured materials to colourless products while being reduced to the chloride ion. It will also oxidize sulfur dioxide to the sulfate ion.
(i) Describe the colour change that occurs when aqueous chlorine is added to aqueous sodium bromide.
(ii) Outline, with the help of a chemical equation, why this reaction occurs.
The colour change in the reaction between aqueous chlorine and aqueous sodium iodide is very similar, but it differs with an excess of aqueous chlorine. Describe the appearance of the reaction mixture when excess aqueous chlorine has been added to aqueous sodium iodide.
Chloric(I) acid is a weak acid, but hydrochloric acid is a strong acid. Outline how this is indicated in the equation above.
State a balanced equation for the reaction of chloric(I) acid with water.
Outline, in terms of the equilibrium above, why it is dangerous to use an acidic toilet cleaner in combination with this kind of bleach.
Suggest why a covalent molecule, such as chloric(I) acid, is readily soluble in water.
Draw the Lewis (electron dot) structure of chloric(I) acid.
Predict the H–O–Cl bond angle in this molecule and explain this in terms of the valence shell electron pair repulsion (VSEPR) theory.
(i) Deduce the coefficients required to balance the half-equations given below.
___ \({\text{Cl}}{{\text{O}}^ - } + \) ___ \({{\text{H}}^ + } + \) ___ \({{\text{e}}^ - } \rightleftharpoons \) ___ \({{\text{H}}_2}{\text{O}} + \) ___ \({\text{C}}{{\text{l}}^ - }\)
___ \({\text{SO}}_4^{2 - }\) ___ \({{\text{H}}^ + } + \) ___ \({{\text{e}}^ - } \rightleftharpoons \) ___ \({\text{S}}{{\text{O}}_2} + \) ___ \({{\text{H}}_2}{\text{O}}\)
(ii) State the initial and final oxidation numbers of both chlorine and sulfur in the equations in part (i).

(iii) Use the half-equations to deduce the balanced equation for the reaction between the chlorate(I) ion and sulfur dioxide.
Markscheme
(i) from (pale) green/colourless to yellow/orange/brown;
Initial colour must be stated.
Do not accept “clear/transparent” instead of “colourless”.
(ii) chlorine more reactive/more powerful oxidizing agent (than bromine);
Accept opposite statements for bromine.
Accept “chloride ion a weaker reducing agent” / “bromide ion a stronger reducing agent”.
Accept “chlorine more electronegative than bromine”.
\({\text{C}}{{\text{l}}_2}{\text{(aq)}} + {\text{2NaBr(aq)}} \to {\text{B}}{{\text{r}}_2}{\text{(aq)}} + {\text{2NaCl(aq)}}\) /
\({\text{C}}{{\text{l}}_2}{\text{(aq)}} + {\text{2B}}{{\text{r}}^ - }{\text{(aq)}} \to {\text{B}}{{\text{r}}_2}{\text{(aq)}} + {\text{2C}}{{\text{l}}^ - }{\text{(aq)}}\);
Ignore state symbols.
Do not accept with equilibrium sign.
solid (in a colourless solution);
Accept “dark brown solution”.
chloric(I) acid (shown as) a molecule/molecular, but hydrochloric acid (shown as being) split into ions / OWTTE;
Accept “chloric(I) acid is partially dissociated and hydrochloric acid is fully dissociated”.
Reference needed to both acids for mark.
\({\text{HOCl(aq)}} \rightleftharpoons {{\text{H}}^ + }{\text{(aq)}} + {\text{Cl}}{{\text{O}}^ - }{\text{(aq)}}/{\text{HOCl(aq)}} + {{\text{H}}_2}{\text{O(l)}} \rightleftharpoons {{\text{H}}_3}{{\text{O}}^ + }{\text{(aq)}} + {\text{Cl}}{{\text{O}}^ - }{\text{(aq)}}\);
Equilibrium sign required for the mark.
Ignore state symbols.
acid displaces the equilibrium to the left (to form chlorine);
chlorine is toxic/poisonous/harmful/lung irritant;
Accept answers that refer to the (c) (ii) equilibrium.
chloric(I) acid has –OH group / hydrogen attached to a very electronegative atom;
Accept polar molecule.
can form hydrogen bonds to water;
hydrogen bonding to water increases its solubility;
(as a weak acid it is) in equilibrium with ions;
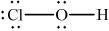 ;
;
Accept lines, dots or crosses to represent electron pairs.
\( \sim\)104°;
Accept values between 102° and 106°.
four electron pairs/regions of high electron density around O atom / electron pairs/regions of high electron density tetrahedrally arranged and two lone/non-bonding electron pairs on O atom;
Accept Lewis structure with two lone pairs on O and two angular bond pairs if given here as equivalent to M2.
lone pair–bonding pair repulsion greater than bonding pair–bonding pair repulsion;
(i) \({\text{(1) Cl}}{{\text{O}}^ - } + \) 2\(\,{{\text{H}}^ + } + \) 2\(\,{{\text{e}}^ - } \rightleftharpoons {\text{(1) }}{{\text{H}}_2}{\text{O}} + {\text{(1) C}}{{\text{l}}^ - }\);
\({\text{(1) SO}}_4^{2 - } + \) 4\(\,{{\text{H}}^ + } + \) 2\(\,{{\text{e}}^ - } \rightleftharpoons {\text{(1) S}}{{\text{O}}_2} + \) 2\(\,{{\text{H}}_2}{\text{O}}\);
(ii) Award [2] for all correct, [1] for 2 or 3 correct.

Remember to apply ECF from previous equations.
Penalize incorrect notation (eg, 4 or 4+ rather than +4) once only, so award [1] for a fully correct answer in an incorrect format.
(iii) \({\text{Cl}}{{\text{O}}^ - }{\text{(aq)}} + {\text{S}}{{\text{O}}_2}{\text{(aq)}} + {{\text{H}}_2}{\text{O(l)}} \rightleftharpoons {\text{SO}}_4^{2 - }{\text{(aq)}} + {\text{2}}{{\text{H}}^ + }{\text{(aq)}} + {\text{C}}{{\text{l}}^ - }{\text{(aq)}}\)
correct reactants and products;
balancing and cancelling \({{\text{e}}^ - }\), \({{\text{H}}^ + }\) and \({{\text{H}}_{\text{2}}}{\text{O}}\);
Ignore state symbols.
Do not penalize equilibrium sign.
Examiners report
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
This was the least popular and the least successfully answered question on the paper. Many were unable to describe the colour change required in (a)(i) though more could give an appropriate equation and explain why the reaction occurred in terms of electronegativity. (b) was essentially a “dead” mark and perhaps was out of place on a SL paper. Many students seemed to be aware of the difference between strong and weak acids, but few could use this to answer (c)(i), and many were unable to write an equation for its reaction in water. The more able candidates realised that acids would affect the position of the equilibrium and a number recognized that the toxic gas chlorine would be a product. Many students identified hydrogen bonding from the –OH group as being the reason for the solubility of HOCl. Most were able to give the Lewis (electron dot) structure of chloric(I) acid, but few were able to give a detailed explanation of its bond angle, with only a minority referring to electron domains. In part (d) very few students could write, or combine, appropriate half equations, even though the reactants and products were given, though many could deduce the oxidation numbers of the species in the equations. Some marks were unfortunately lost as candidates omitted the sign.
Ethane-1,2-diol, HOCH2CH2OH, has a wide variety of uses including the removal of ice from aircraft and heat transfer in a solar cell.
Ethane-1,2-diol can be formed according to the following reaction.
2CO (g) + 3H2 (g) \( \rightleftharpoons \) HOCH2CH2OH (g)
(i) Deduce the equilibrium constant expression, Kc, for this reaction.
(ii) State how increasing the pressure of the reaction mixture at constant temperature will affect the position of equilibrium and the value of Kc.
Position of equilibrium:
Kc:
(iii) Calculate the enthalpy change, ΔHθ, in kJ, for this reaction using section 11 of the data booklet. The bond enthalpy of the carbon–oxygen bond in CO (g) is 1077kJmol-1.
(iv) The enthalpy change, ΔHθ, for the following similar reaction is –233.8 kJ.
2CO(g) + 3H2(g) \( \rightleftharpoons \) HOCH2CH2OH (l)
Deduce why this value differs from your answer to (a)(iii).
Determine the average oxidation state of carbon in ethene and in ethane-1,2-diol.
Ethene:
Ethane-1,2-diol:
Explain why the boiling point of ethane-1,2-diol is significantly greater than that of ethene.
Ethane-1,2-diol can be oxidized first to ethanedioic acid, (COOH)2, and then to carbon dioxide and water. Suggest the reagents to oxidize ethane-1,2-diol.
Markscheme
(i)
\(\ll {K_{\text{C}}} = \gg \frac{{\left[ {{\text{HOC}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{OH}}} \right]}}{{{{\left[ {{\text{CO}}} \right]}^{\text{2}}} \times {{\left[ {{{\text{H}}_{\text{2}}}} \right]}^{\text{3}}}}}\)
(ii)
Position of equilibrium: moves to right OR favours product
Kc: no change OR is a constant at constant temperature
(iii)
Bonds broken: 2C≡O + 3(H-H) / 2(1077kJmol-1) + 3(436kJmol-1) / 3462 «kJ»
Bonds formed: 2(C-O) + 2(O-H) + 4(C-H) + (C-C) / 2(358kJmol-1) + 2(463kJmol-1) + 4(414kJmol-1) + 346kJmol-1 / 3644 «kJ»
«Enthalpy change = bonds broken - bonds formed = 3462 kJ - 3644 kJ =» -182 «kJ»
Award [3] for correct final answer.
Award [2 max] for «+»182 «kJ».
(iv)
in (a)(iii) gas is formed and in (a)(iv) liquid is formed
OR
products are in different states
OR
conversion of gas to liquid is exothermic
OR
conversion of liquid to gas is endothermic
OR
enthalpy of vapourisation needs to be taken into account
Accept product is «now» a liquid.
Accept answers referring to bond enthalpies being means/averages.
Ethene: –2
Ethane-1,2-diol: –1
Do not accept 2–, 1– respectively.
ethane-1,2-diol can hydrogen bond to other molecules «and ethene cannot»
OR
ethane-1,2-diol has «significantly» greater van der Waals forces
Accept converse arguments.
Award [0] if answer implies covalent bonds are broken
hydrogen bonding is «significantly» stronger than other intermolecular forces
acidified «potassium» dichromate«(VI)»/H+ AND K2Cr2O7/H+ AND Cr2O72-
OR
«acidified potassium» manganate(VII)/ «H+» KMnO4 /«H+» MnO4-
Accept Accept H2SO4 or H3PO4 for H+.
Accept “permanganate” for “manganate(VII)”.
Examiners report
Consider the following equilibrium:
\[\begin{array}{*{20}{l}} {{\text{4N}}{{\text{H}}_3}{\text{(g)}} + {\text{5}}{{\text{O}}_2}{\text{(g)}} \rightleftharpoons {\text{4NO(g)}} + {\text{6}}{{\text{H}}_2}{\text{O(g)}}}&{\Delta {H^\Theta } = - 909{\text{ kJ}}} \end{array}\]
Nitrogen reacts with hydrogen to form ammonia in the Haber process, according to the following equilibrium.
\[\begin{array}{*{20}{l}} {{{\text{N}}_{\text{2}}}{\text{(g)}} + {\text{3}}{{\text{H}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{2N}}{{\text{H}}_{\text{3}}}{\text{(g)}}}&{\Delta {H^\Theta } = - 92.6{\text{ kJ}}} \end{array}\]
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for the reaction.
Predict the direction in which the equilibrium will shift when the following changes occur.
The volume increases.
The temperature decreases.
\({{\text{H}}_{\text{2}}}{\text{O(g)}}\) is removed from the system.
A catalyst is added to the reaction mixture.
Define the term activation energy, \({E_{\text{a}}}\).
Nitrogen monoxide, NO, is involved in the decomposition of ozone according to the following mechanism.
\[\begin{array}{*{20}{l}} {}&{{{\text{O}}_{\text{3}}} \to {{\text{O}}_{\text{2}}} + {\text{O}} \bullet } \\ {}&{{{\text{O}}_3} + {\text{NO}} \to {\text{N}}{{\text{O}}_2} + {{\text{O}}_2}} \\ {}&{{\text{N}}{{\text{O}}_2} + {\text{O}} \bullet \to {\text{NO}} + {{\text{O}}_2}} \\ {{\text{Overall:}}}&{{\text{2}}{{\text{O}}_3} \to {\text{3}}{{\text{O}}_2}} \end{array}\]
State and explain whether or not NO is acting as a catalyst.
Define the term endothermic reaction.
Sketch the Maxwell-Boltzmann energy distribution curve for a reaction with and without a catalyst, and label both axes.
Define the term rate of reaction.
Iron, used as the catalyst in the Haber process, has a specific heat capacity of \({\text{0.4490 J}}\,{{\text{g}}^{ - 1}}{{\text{K}}^{ - 1}}\). If 245.0 kJ of heat is supplied to 8.500 kg of iron, initially at a temperature of 15.25 °C, determine its final temperature in K.
Markscheme
\({\text{(}}{K_{\text{c}}}{\text{)}} = \frac{{{{{\text{[NO]}}}^4}{{{\text{[}}{{\text{H}}_2}{\text{O]}}}^6}}}{{{{{\text{[N}}{{\text{H}}_3}{\text{]}}}^4}{{{\text{[}}{{\text{O}}_2}{\text{]}}}^5}}}\);
No mark if square brackets are omitted or are incorrect.
right;
right;
right;
no change;
minimum energy needed (by reactants/colliding particles) to react/start/initiate a reaction;
Allow energy difference between reactants and transition state.
catalyst;
regenerated at end of reaction / OWTTE;
(system) absorbs/takes in heat from surroundings / OWTTE;
Allow standard enthalpy change/ \(\Delta {H^\Theta }\) positive.
Allow bond breaking more energetic then bond formation / OWTTE.
Absorbs/takes in heat alone not sufficient for mark.
Curve showing:
general shape of Maxwell-Boltzmann energy distribution curve;
correct position of \({E_{\text{a}}}\) (catalysed) and \({E_{\text{a}}}\) (uncatalysed);
labelled y-axis: probability of particles (with that kinetic energy) and labelled x-axis: (kinetic) energy;
Allow number/fraction/proportion of particles (with kinetic energy) for y-axis label, but do not allow amount or particles.

Award [2 max] if a second curve is drawn, but at a higher temperature, M2 will not be scored here.
change in concentration of reactant/product with time / rate of change of concentration;
Increase can be used instead of change for product or decrease can be used instead of change for reactant.
Allow mass/amount/volume instead of concentration.
Do not accept substance.
\(q = mc\Delta T = 2.450 \times {10^5} = (8.500 \times {10^3})(0.4490)({T_{\text{f}}} - 15.25)\);
\({T_{\text{f}}} = 79.44{\text{ °C}}/\Delta T = 64.19{\text{ (°C}}/{\text{K)}}\);
\({T_{\text{f}}} = (79.44 + 273) = 352{\text{ (K)}}\);
Award [3] for correct final answer.
Accept the use of 273.15 K instead of 273 K giving final value of 352.59 K.
For M1 and M2 award [1 max] for use of \(q = mc\Delta T\) if incorrect units of m and c are used.
Examiners report
In part (a) of this question the \({K_{\text{c}}}\) expression was usually written correctly though the very weak students did mix up the numerator and denominator in (i), or include a + sign between substances.
Candidates generally had few problems, but the reaction condition that proved to be the most the most difficult factor was the volume.
Activation energy was often clearly defined though some forgot to mention minimum.
The best students realised that NO acted as a catalyst as it was regenerated at the end of the reaction. However many weaker students stated it was not a catalyst as it was not involved in the reaction.
The definition of an endothermic reaction was generally well answered, however some just said it absorbs heat and forgot to mention the surroundings in their answer.
Incorrect labels for the axes were often seen, as well as a very high proportion of symmetrical curves, some which did not start at the origin. Also many drew two curves. Also in some cases the catalyzed and uncatalyzed activation energies were often mixed up. The weaker students drew an enthalpy level diagram instead of a Maxwell-Boltzmann distribution.
In the definition for rate of reaction some students forgot to mention concentration.
Consider the following three redox reactions.
\[\begin{array}{*{20}{l}} {{\text{Cd(s)}} + {\text{N}}{{\text{i}}^{2 + }}{\text{(aq)}} \to {\text{C}}{{\text{d}}^{2 + }}{\text{(aq)}} + {\text{Ni(s)}}} \\ {{\text{Ni(s)}} + {\text{2A}}{{\text{g}}^ + }{\text{(aq)}} \to {\text{N}}{{\text{i}}^{2 + }}{\text{(aq)}} + {\text{2Ag(s)}}} \\ {{\text{Zn(s)}} + {\text{C}}{{\text{d}}^{2 + }}{\text{(aq)}} \to {\text{Z}}{{\text{n}}^{2 + }}{\text{(aq)}} + {\text{Cd(s)}}} \end{array}\]
(i) Draw an annotated diagram of a voltaic cell composed of a magnesium electrode in \({\text{1.0 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) magnesium nitrate solution and a silver electrode in \({\text{1.0 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) silver nitrate solution. State the direction of electron flow on your diagram.
(ii) Deduce half-equations for the oxidation and reduction reactions.
(i) Deduce the order of reactivity of the four metals, cadmium, nickel, silver and zinc and list in order of decreasing reactivity.
(ii) Identify the best oxidizing agent and the best reducing agent.
(i) Solid sodium chloride does not conduct electricity but molten sodium chloride does. Explain this difference.
(ii) Outline what happens in an electrolytic cell during the electrolysis of molten sodium chloride using inert electrodes. Deduce equations for the reactions occurring at each electrode.
(i) A state of equilibrium can exist when a piece of copper metal is placed in a solution of copper(II) sulfate. Outline the characteristics of a chemical system in dynamic equilibrium.
(ii) For an exothermic reaction state how an increase in temperature would affect both \({K_{\text{c}}}\) and the position of equilibrium.
Markscheme
(i) 
correctly labelled electrodes and solutions;
labelled salt bridge;
voltmeter;
Allow bulb or ammeter.
direction of electron flow;
(ii) Oxidation:
\({\text{Mg(s)}} \to {\text{M}}{{\text{g}}^{2 + }}{\text{(aq)}} + {\text{2}}{{\text{e}}^ - }\);
Reduction:
\({\text{A}}{{\text{g}}^ + }{\text{(aq)}} + {{\text{e}}^ - } \to {\text{Ag(s)}}\);
Ignore state symbols.
Award [1 max] if equations not labelled reduction or oxidation or labelled the wrong way round.
Allow e instead of e–.
Penalize equilibrium sign or reversible arrows once only in parts (a) (ii) and (c) (ii).
(i) \({\text{Zn}} > {\text{Cd}} > {\text{Ni}} > {\text{Ag}}\)
Zn most reactive;
rest of order correct;
(ii) Best oxidizing agent:
\({\text{A}}{{\text{g}}^ + }\);
Do not accept Ag.
Best reducing agent:
Zn;
Do not accept Zn2+.
(i) sodium chloride crystals consist of ions in a (rigid) lattice / ions cannot move (to electrodes) / OWTTE;
when melted ions free to move / ions move when potential difference/voltage applied;
(ii) positive sodium ions/\({\text{N}}{{\text{a}}^ + }\) move to negative electrode/cathode and negative chloride ions/ \({\text{C}}{{\text{l}}^ - }\) move to positive electrode/anode;
electrons released to positive electrode/anode by negative ions and accepted from negative electrode/cathode by positive ions / reduction occurs at the negative electrode/cathode and oxidation occurs at the positive electrode/anode / \({\text{N}}{{\text{a}}^ + }\) ions are reduced and \({\text{C}}{{\text{l}}^ - }\) ions are oxidized;
(Positive electrode/anode):
\({\text{2C}}{{\text{l}}^ - } \to {\text{C}}{{\text{l}}_2} + {\text{2}}{{\text{e}}^ - }{\text{ / C}}{{\text{l}}^ - } \to \frac{1}{2}{\text{C}}{{\text{l}}_2} + {{\text{e}}^ - }\);
(Negative electrode/cathode):
\({\text{2N}}{{\text{a}}^ + } + {\text{2}}{{\text{e}}^ - } \to {\text{2Na / N}}{{\text{a}}^ + } + {{\text{e}}^ - } \to {\text{Na}}\);
Award [1 max] if equations not labelled or labelled wrong way round.
Allow e instead of e–.
Penalize equilibrium sign or reversible arrows once only in parts (a) (ii) and (c) (ii).
(i) macroscopic properties remain constant / concentrations remain constant / no change to copper solution seen;
rate of reverse/backwards reaction = rate of forward reaction;
(ii) \({K_{\text{c}}}\) decreases;
position of equilibrium shifts to left;
Examiners report
This question was also chosen by approximately 40% of candidates. In (a), the diagram was reasonably attempted by most candidates, with just a few candidates giving both electrodes in one beaker. Some candidates omitted to include a voltmeter and other common mistakes included omission of states and incorrect direction of electron flow. In (ii), fewer candidates scored these marks and many equations were not labelled explicitly as oxidation and reduction. Other common errors included incorrect charges for the silver and magnesium ions.
In (b), most candidates were able to place the metals in order, though a small minority misread the question, and gave zinc as the least reactive. In (ii), zinc was generally given as the best reducing agent, but often silver metal rather than silver(I) ion as given as the best oxidizing agent.
There were many references to sodium chloride having a metallic structure in (c) (i), and describing its conduction in terms of electrons rather than ions. In (ii), very few candidates mentioned that the ions move towards the oppositely charged electrode. The nature of the electrolytic process was not well explained.
The characteristics of a chemical system in a dynamic equilibrium in (d) (i) typically were understood by most candidates, although many just scored one mark. (ii) was well answered.
Consider the following reaction taking place at 375 °C in a \({\text{1.00 d}}{{\text{m}}^{\text{3}}}\) closed container.
\[\begin{array}{*{20}{l}} {{\text{C}}{{\text{l}}_{\text{2}}}{\text{(g)}} + {\text{S}}{{\text{O}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{S}}{{\text{O}}_{\text{2}}}{\text{C}}{{\text{l}}_{\text{2}}}{\text{(g)}}}&{\Delta {H^\Theta } = - 84.5{\text{ kJ}}} \end{array}\]
A solution of hydrogen peroxide, \({{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}\), is added to a solution of sodium iodide, NaI, acidified with hydrochloric acid, HCl. The yellow colour of the iodine, \({{\text{I}}_{\text{2}}}\), can be used to determine the rate of reaction.
\[{{\text{H}}_2}{{\text{O}}_2}{\text{(aq)}} + {\text{2NaI(aq)}} + {\text{2HCl(aq)}} \to {\text{2NaCl(aq)}} + {{\text{I}}_2}{\text{(aq)}} + {\text{2}}{{\text{H}}_2}{\text{O(l)}}\]
The experiment is repeated with some changes to the reaction conditions. For each of the changes that follow, predict, stating a reason, its effect on the rate of reaction.
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for the reaction.
If the temperature of the reaction is changed to 300 °C, predict, stating a reason in each case, whether the equilibrium concentration of \({\text{S}}{{\text{O}}_{\text{2}}}{\text{C}}{{\text{l}}_{\text{2}}}\) and the value of \({K_{\text{c}}}\) will increase or decrease.
If the volume of the container is changed to \({\text{1.50 d}}{{\text{m}}^{\text{3}}}\), predict, stating a reason in each case, how this will affect the equilibrium concentration of \({\text{S}}{{\text{O}}_{\text{2}}}{\text{C}}{{\text{l}}_{\text{2}}}\) and the value of \({K_{\text{c}}}\).
Suggest, stating a reason, how the addition of a catalyst at constant pressure and temperature will affect the equilibrium concentration of \({\text{S}}{{\text{O}}_{\text{2}}}{\text{C}}{{\text{l}}_{\text{2}}}\).
Graphing is an important method in the study of the rates of chemical reaction. Sketch a graph to show how the reactant concentration changes with time in a typical chemical reaction taking place in solution. Show how the rate of the reaction at a particular time can be determined.
The concentration of \({{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}\) is increased at constant temperature.
The solution of NaI is prepared from a fine powder instead of large crystals.
Explain why the rate of a reaction increases when the temperature of the system increases.
Markscheme
\(({K_{\text{c}}}) = \frac{{{\text{[S}}{{\text{O}}_2}{\text{C}}{{\text{l}}_2}{\text{]}}}}{{{\text{[C}}{{\text{l}}_2}{\text{][S}}{{\text{O}}_2}{\text{]}}}}\);
Ignore state symbols.
Square brackets [ ] required for the equilibrium expression.
value of \({K_{\text{c}}}\) increases;
\({\text{[S}}{{\text{O}}_2}{\text{C}}{{\text{l}}_2}{\text{]}}\) increases;
decrease in temperature favours (forward) reaction which is exothermic;
Do not allow ECF.
no effect on the value of \({K_{\text{c}}}\) / depends only on temperature;
\({\text{[S}}{{\text{O}}_2}{\text{C}}{{\text{l}}_2}{\text{]}}\) decreases;
increase in volume favours the reverse reaction which has more gaseous moles;
Do not allow ECF.
no effect;
catalyst increases the rate of forward and reverse reactions (equally) / catalyst decreases activation energies (equally);

labelled axes (including appropriate units);
correctly drawn curve;
correctly drawn tangent;
rate equal to slope/gradient of tangent (at given time) / rate \( = \frac{y}{x}\) at time \(t\);
[3 max] for straight line graph or graph showing product formation.
increases rate of reaction;
molecules (of \({{\text{H}}_2}{{\text{O}}_2}\)) collide more frequently / more collisions per unit time;
No ECF here.
no effect / (solution) remains unchanged;
solid NaI is not reacting / aqueous solution of NaI is reacting / surface area of NaI is not relevant in preparing the solution / OWTTE;
kinetic energy/speed of reacting molecules increases;
frequency of collisions increases per unit time;
greater proportion of molecules have energy greater than activation energy/\({E_{\text{a}}}\);
Accept more energetic collisions.
Examiners report
This was the most popular question in Section B and there was a generally pleasing level of performance. In (a)(i) most candidates were able to correctly deduce the equilibrium constant.
In (ii) most candidates realized the exothermic reaction would be favoured, and gained full marks for their explanation. However, some candidates seemed not to appreciate that the specified temperature of 300 °C was lower than the original, and so based their answers on a temperature increase.
In (iii) most forgot to mention the word gaseous when talking about the particles and many forgot that \({K_{\text{c}}}\) is only affected by temperature.
In (iv) candidates correctly stated that concentration would not change and stated that reaction rates of both forward and reverse reactions would be affected equally. However, some answered ‘the addition of a catalyst does not affect \({K_{\text{c}}}\) or the position of equilibrium’ which did not answer the question and scored no marks as they had not commented on the concentration of \({\text{SOC}}{{\text{l}}_{\text{2}}}\).
For (b), although most students were able to correctly sketch the reactant concentration / time graph by labeling the axes and drawing an appropriate curve, some candidates incorrectly read the question and sketched product / concentration time curve. Drawing a tangent to determine the rate was not well known and only some were able to describe how the rate at a particular instant could be determined from the tangent to the curve.
In (c), most scored the marks in (i) and were able to correctly describe the effect of concentration on rate in terms of collision theory, although some forgot to mention the frequency of the collisions just stating there would be more.
In part (ii), most candidates assumed that the rate would increase with surface area of the solute, and few realized that once the sodium iodide was in solution then the particle size of the solid used to make it was not relevant as it is the solution which reacts.
Part (d) was well answered but some candidates lost marks due to imprecise responses. For example it is the kinetic energy that increases with temperature, not energy. Also there were some errors such as the omission of the idea of frequency when referring to collisions and the belief that an increase in temperature caused a decrease in activation energy.
An equilibrium occurs between nitrogen dioxide, \({\text{N}}{{\text{O}}_{\text{2}}}{\text{(g)}}\), and dinitrogen tetroxide, \({{\text{N}}_{\text{2}}}{{\text{O}}_{\text{4}}}{\text{(g)}}\).
\({\text{2N}}{{\text{O}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {{\text{N}}_{\text{2}}}{{\text{O}}_{\text{4}}}{\text{(g)}}\) \(\Delta H = - 58{\text{ kJ}}\)
Deduce the equilibrium constant expression, \({K_{\text{c}}}\), for this reaction.
Explain the effect on the position of the equilibrium and on the value of \({K_{\text{c}}}\) when:
(i) pressure is decreased and temperature is kept constant.
(ii) temperature is increased and pressure is kept constant.
Markscheme
\(({K_{\text{c}}} = )\frac{{{\text{[}}{{\text{N}}_2}{{\text{O}}_4}{\text{(g)]}}}}{{{{{\text{[N}}{{\text{O}}_2}{\text{(g)]}}}^2}}}\);
Ignore state symbols.
(i) equilibrium shifts to left as there are more moles (of gas) on reactant side;
no change to \({K_{\text{c}}}\) as it is a constant at fixed temperature / OWTTE;
Award [1 max] for correct equilibrium shift and \({K_c}\) change if no explanation given.
(ii) equilibrium shifts to left since reaction is exothermic/\(\Delta H\) negative / reverse reaction is endothermic/absorbs heat;
value of \({K_{\text{c}}}\) decreases because less \({{\text{N}}_2}{{\text{O}}_4}\) and more \({\text{N}}{{\text{O}}_2}\) / OWTTE;
Award [1 max] for correct equilibrium shitft and \({K_c}\) change if no explanation given.
Examiners report
The equilibrium constant expression for the equilibrium between \({\text{N}}{{\text{O}}_{\text{2}}}\) and \({{\text{N}}_{\text{2}}}{{\text{O}}_{\text{4}}}\) was well done by most candidates in 3(a) although a few had the expression upside down and several separated the molecules into atoms. Students generally could state and explain the shift in equilibrium position but had more trouble with the effect on the equilibrium constant in both (b)(i) and (b)(ii). It was rare to find a response that stated the changes and explained them for both the equilibrium position and the equilibrium constant. Candidates should take care to answer all parts of the question.
The equilibrium constant expression for the equilibrium between \({\text{N}}{{\text{O}}_{\text{2}}}\) and \({{\text{N}}_{\text{2}}}{{\text{O}}_{\text{4}}}\) was well done by most candidates in 3(a) although a few had the expression upside down and several separated the molecules into atoms. Students generally could state and explain the shift in equilibrium position but had more trouble with the effect on the equilibrium constant in both (b)(i) and (b)(ii). It was rare to find a response that stated the changes and explained them for both the equilibrium position and the equilibrium constant. Candidates should take care to answer all parts of the question.
The Haber process enables the large-scale production of ammonia needed to make fertilizers.
The equation for the Haber process is given below.
\[{{\text{N}}_2}({\text{g)}} + 3{{\text{H}}_2}({\text{g)}} \rightleftharpoons {\text{2N}}{{\text{H}}_3}({\text{g)}}\]
The percentage of ammonia in the equilibrium mixture varies with temperature.

Fertilizers may cause health problems for babies because nitrates can change into nitrites in water used for drinking.
A student decided to investigate the reactions of the two acids with separate samples of \({\text{0.20 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) sodium hydroxide solution.
(i) Use the graph to deduce whether the forward reaction is exothermic or endothermic and explain your choice.
(ii) State and explain the effect of increasing the pressure on the yield of ammonia.
(iii) Explain the effect of increasing the temperature on the rate of reaction.
(i) Define oxidation in terms of oxidation numbers.
(ii) Deduce the oxidation states of nitrogen in the nitrate, \({\text{NO}}_{\text{3}}^ - \), and nitrite, \({\text{NO}}_{\text{2}}^ - \), ions.
The nitrite ion is present in nitrous acid, HNO2, which is a weak acid. The nitrate ion is present in nitric acid, HNO3, which is a strong acid. Distinguish between the terms strong and weak acid and state the equations used to show the dissociation of each acid in aqueous solution.
A small piece of magnesium ribbon is added to solutions of nitric and nitrous acid of the same concentration at the same temperature. Describe two observations that would allow you to distinguish between the two acids.
(i) Calculate the volume of the sodium hydroxide solution required to react exactly with a \({\text{15.0 c}}{{\text{m}}^{\text{3}}}\) solution of \({\text{0.10 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) nitric acid.
(ii) The following hypothesis was suggested by the student: “Since nitrous acid is a weak acid it will react with a smaller volume of the \({\text{0.20 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) sodium hydroxide solution.” Comment on whether or not this is a valid hypothesis.
The graph below shows how the conductivity of the two acids changes with concentration.

Identify Acid 1 and explain your choice.
Nitric acid reacts with silver in a redox reaction.
__ \({\text{Ag(s)}} + \) __ \({\text{NO}}_3^ - {\text{(aq)}} + \) ___ \( \to \) ___\({\text{A}}{{\text{g}}^ + }{\text{(aq)}} + \) __ \({\text{NO(g)}} + \) ____
Using oxidation numbers, deduce the complete balanced equation for the reaction showing all the reactants and products.
Markscheme
(i) exothermic;
Accept either of the following for the second mark.
increasing temperature favours endothermic/reverse reaction;
as yield decreases with increasing temperature;
(ii) yield increases / equilibrium moves to the right / more ammonia;
increase in pressure favours the reaction which has fewer moles of gaseous products;
(iii) (rate increases because) increase in frequency (of collisions);
increase in energy (of collisions);
more colliding molecules with \(E \geqslant {E_{\text{a}}}\);
(i) increase in the oxidation number;
(ii) (NO3) + 5 and (NO2–) + 3;
Accept V and III.
Do not penalize missing charges on numbers.
strong acid completely dissociated/ionized and weak acid partially dissociated/ionized;
\({\text{HN}}{{\text{O}}_3}{\text{(aq)}} \to {{\text{H}}^ + }{\text{(aq)}} + {\text{NO}}_3^ - {\text{(aq)}}\);
\({\text{HN}}{{\text{O}}_2}{\text{(aq)}} \rightleftharpoons {{\text{H}}^ + }{\text{(aq)}} + {\text{NO}}_2^ - {\text{(aq)}}\);
Allow only arrows as shown.
State symbols not needed.
Accept H2O and H3O+.
With HNO3:
faster rate of bubble/gas/hydrogen production;
faster rate of magnesium dissolving;
higher temperature change;
Accept opposite argument for HNO2.
Award [1] if 2 observations given but acid is not identified.
Reference to specific observations needed.
(i) (nitric acid) \({\text{7.5 c}}{{\text{m}}^{\text{3}}}\);
(ii) not valid as nitrous acid reacts with same volume/ \({\text{7.5 c}}{{\text{m}}^{\text{3}}}\);
HNO3;
(higher conductivity for solutions with same concentration as) there are more ions in solution;
change in oxidation numbers: Ag from 0 to +1 and N from +5 to +2;
Do not penalise missing charges on numbers.
balanced equation: \({\text{3Ag}} + {\text{NO}}_3^ - + {\text{4}}{{\text{H}}^ + } \to {\text{3A}}{{\text{g}}^ + } + {\text{NO}} + {\text{2}}{{\text{H}}_2}{\text{O}}\)
Award [1] for correct reactants and product;
Award [3] for correct balanced equation.
Ignore state symbols
Examiners report
This was the most popular question and it was well answered by the majority of candidates. The reaction was correctly described as exothermic and the reason for this explained correctly in most cases. Most candidates knew that yield would increase with increased pressure, but failed to score a second mark because they did not mention ‘gaseous’ although they did know the answer. The effect of increased temperature on rate was generally well described although some did get confused with yield and how it would affect equilibrium.
Most candidates correctly defined oxidation in 6(b)(i) but ‘hedged their bets’ by stating loss of electrons as well as an increase in oxidation number. In 6(b)(ii) the oxidation states were generally deduced correctly but sometimes written as ionic charges (5+ for instance, instead of +5).
In 6(c) most correctly defined strong and weak acids, and many also wrote correct equations. A few, though, had no idea. In (c), arrows proved to be a minefield for several candidates, especially the equilibrium sign. HA was commonly given, as were CH3COOH and HCl, instead of nitric and nitrous acid.
6(d) presented problems with many candidates unable to describe observations and instead stating there would be ‘more hydrogen produced’ or just that ‘the reaction would be faster’. However, better candidates were able to answer this part correctly and scored full marks.
In 6(e)(i) the calculation was answered well, but 6(e)(ii), that asked for a comment on the hypothesis, was not and few candidates stated that the same volume of acid was needed.
In 6(f), the majority correctly identified the strong acid but often failed to explain its better conductivity in terms of the ions.
Many could give a correct balanced equation and scored the 3 marks, and others scored 1 mark for giving the correct reactants and products. However, not many candidates used oxidation numbers to deduce the balanced equation.
A class studied the equilibrium established when ethanoic acid and ethanol react together in the presence of a strong acid, using propanone as an inert solvent. The equation is given below.
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{COOH}} + {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{OH}} \rightleftharpoons {\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}} + {{\text{H}}_{\text{2}}}{\text{O}}\]
One group made the following initial mixture:

After one week, a \(5.00 \pm 0.05{\text{ c}}{{\text{m}}^{\text{3}}}\) sample of the final equilibrium mixture was pipetted out and titrated with \({\text{0.200 mol}}\,{\text{d}}{{\text{m}}^{ - 2}}\) aqueous sodium hydroxide to determine the amount of ethanoic acid remaining. The following titration results were obtained:

The density of ethanoic acid is \({\text{1.05 g}}\,{\text{c}}{{\text{m}}^{ - 3}}\). Determine the amount, in mol, of ethanoic acid present in the initial mixture.
The hydrochloric acid does not appear in the balanced equation for the reaction. State its function.
Identify the liquid whose volume has the greatest percentage uncertainty.
(i) Calculate the absolute uncertainty of the titre for Titration 1 (\({\text{27.60 c}}{{\text{m}}^{\text{3}}}\)).
(ii) Suggest the average volume of alkali, required to neutralize the \({\text{5.00 c}}{{\text{m}}^{\text{3}}}\) sample, that the student should use.
(iii) \({\text{23.00 c}}{{\text{m}}^{\text{3}}}\) of this \({\text{0.200 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) aqueous sodium hydroxide reacted with the ethanoic acid in the \({\text{5.00 c}}{{\text{m}}^{\text{3}}}\) sample. Determine the amount, in mol, of ethanoic acid present in the \({\text{50.0 c}}{{\text{m}}^{\text{3}}}\) of final equilibrium mixture.
Referring back to your answer for part (a), calculate the percentage of ethanoic acid converted to ethyl ethanoate.
Deduce the equilibrium constant expression for the reaction.
Outline how you could establish that the system had reached equilibrium at the end of one week.
Outline why changing the temperature has only a very small effect on the value of the equilibrium constant for this equilibrium.
Outline how adding some ethyl ethanoate to the initial mixture would affect the amount of ethanoic acid converted to product.
Propanone is used as the solvent because one compound involved in the equilibrium is insoluble in water. Identify this compound and explain why it is insoluble in water.
Suggest one other reason why using water as a solvent would make the experiment less successful.
Markscheme
\({\text{M(C}}{{\text{H}}_3}{\text{COOH)}}\left( { = (4 \times 1.01) + (2 \times 12.01) + (2 \times 16.00)} \right) = 60.06{\text{ (g}}\,{\text{mo}}{{\text{l}}^{ - 1}}{\text{)}}\);
Accept 60 (g mol–1).
\({\text{mass (C}}{{\text{H}}_3}{\text{COOH)}}( = 5.00 \times 1.05) = 5.25{\text{ (g)}}\);
\(\frac{{5.25}}{{{\text{60.06}}}} = 0.0874{\text{ (mol)}}\);
Award [3] for correct final answer.
Accept 0.0875 (comes from using Mr = 60 g mol–1).
catalyst / OWTTE;
hydrochloric acid/HCl;
(i) \( \pm 0.1/0.10{\text{ (c}}{{\text{m}}^3}{\text{)}}\);
Do not accept without ±.
(ii) \({\text{26.00 (c}}{{\text{m}}^3}{\text{)}}\);
(iii) \(0.200 \times \frac{{23.00}}{{1000}} = 0.0046\);
\({\text{0.0046}} \times \frac{{{\text{50.0}}}}{{{\text{5.00}}}} = {\text{0.0460 (mol)}}\);
\(\frac{{0.0874 - 0.0460}}{{0.0874}} \times 100 = 47.4\% \);
\({\text{(}}{K_{\text{c}}} = {\text{)}}\frac{{{\text{[C}}{{\text{H}}_3}{\text{COO}}{{\text{C}}_2}{{\text{H}}_3}{\text{][}}{{\text{H}}_2}{\text{O]}}}}{{{\text{[}}{{\text{C}}_2}{{\text{H}}_5}{\text{OH][C}}{{\text{H}}_3}{\text{COOH]}}}}\);
Do not penalize minor errors in formulas.
Accept \({\text{(}}{K_{\text{c}}} = {\text{)}}\frac{{{\text{[}}ester{\text{][}}water{\text{]}}}}{{{\text{[}}ethanol / alcohol{\text{][(}}ethanoic{\text{)}} acid{\text{]}}}}\).
repeat the titration a day/week later (and result should be the same) / OWTTE;
Accept “concentrations/physical properties/macroscopic properties of the system do not change”.
enthalpy change/\(\Delta H\) for the reaction is (very) small / OWTTE;
decreases (the amount of ethanoic acid converted);
Accept “increases amount of ethanoic acid present at equilibrium” / OWTTE.
(adding product) shifts position of equilibrium towards reactants/LHS / increases the rate of the reverse reaction / OWTTE;
ethyl ethanoate/\({\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}\);
forms only weak hydrogen bonds (to water);
Allow “does not hydrogen bond to water” / “hydrocarbon sections too long” / OWTTE.
M2 can only be given only if M1 correct.
(large excess of) water will shift the position of equilibrium (far to the left) / OWTTE;
Accept any other chemically sound response, such as “dissociation of ethanoic acid would affect equilibrium”.
Examiners report
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
Generally candidates found some elements of this question quite challenging but there were accessible marks of even the weakest candidates. The majority of students were able to determine the molar mass of ethanoic acid but some struggled to calculate the mass from the volume. Most candidates were able to identify the role of hydrochloric acid as a catalyst but some struggled to identify the liquid whose volume had the greatest uncertainty. Most candidates were able to calculate the absolute uncertainty of the titre but some lost a mark by omitting the \( + \)/\( - \) sign. Candidates did not identify the first titre as incongruent and simply averaged the three values which perhaps suggests limited experimental experience. Most students could determine an equilibrium constant expression, but many did not answer the question in (g) and did not suggest how the equilibrium could be established experimentally with many referring to the equal rate of the forward and backward reaction. Many candidates were aware of Le Chatelier effects on the position of equilibrium, but a significant number failed to use this information to answer the question asked and could not explain the small effect of temperature changes. Whilst most students managed to identify the ester as the component of the mixture that was insoluble in water, many did not refer to its inability to form strong hydrogen bonds to water which was necessary for the mark. Quite a number of students came up with a valid reason why water would not be a suitable though some students appeared to have overlooked that the question asked for “one other reason” than that implied in (j).
In acidic solution, ions containing titanium can react according to the half-equation below.
\[{\text{Ti}}{{\text{O}}^{2 + }}{\text{(aq)}} + {\text{2}}{{\text{H}}^ + }{\text{(aq)}} + {{\text{e}}^ - } \rightleftharpoons {\text{T}}{{\text{i}}^{3 + }}{\text{(aq)}} + {{\text{H}}_2}{\text{O(l)}}\]
A reactivity series comparing titanium, cadmium and europium is given below.
Least reactive Cd \( < \) Ti \( < \) Eu Most reactive
The half-equations corresponding to these metals are:
\({\text{E}}{{\text{u}}^{2 + }}{\text{(aq)}} + {\text{2}}{{\text{e}}^ - } \rightleftharpoons {\text{Eu(s)}}\)
\({\text{T}}{{\text{i}}^{3 + }}{\text{(aq)}} + {\text{3}}{{\text{e}}^ - } \rightleftharpoons {\text{Ti(s)}}\)
\({\text{C}}{{\text{d}}^{2 + }}{\text{(aq)}} + {\text{2}}{{\text{e}}^ - } \rightleftharpoons {\text{Cd(s)}}\)
Some students were provided with a \({\text{0.100 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) solution of a monobasic acid, HQ, and given the problem of determining whether HQ was a weak acid or a strong acid.
State the initial and final oxidation numbers of titanium and hence deduce whether it is oxidized or reduced in this change.

Considering the above equilibrium, predict, giving a reason, how adding more acid would affect the strength of the \({\text{Ti}}{{\text{O}}^{2 + }}\) ion as an oxidizing agent.
Deduce which of the species would react with titanium metal.
Deduce the balanced equation for this reaction.
Deduce which of the six species is the strongest oxidizing agent.
A voltaic cell can be constructed using cadmium and europium half-cells. State how the two solutions involved should be connected and outline how this connection works.
Define a Brønsted–Lowry acid.
Distinguish between the terms strong acid and weak acid.
Neelu and Charles decided to solve the problem by determining the volume of \({\text{0.100 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) sodium hydroxide solution needed to neutralize \({\text{25.0 c}}{{\text{m}}^{\text{3}}}\) of the acid. Outline whether this was a good choice.
Neelu and Charles decided to compare the volume of sodium hydroxide solution needed with those required by known \({\text{0.100 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) strong and weak acids. Unfortunately they chose sulfuric acid as the strong acid. Outline why this was an unsuitable choice.
State a suitable choice for both the strong acid and the weak acid.
Strong acid:
Weak acid:
Francisco and Shamiso decided to measure the pH of the initial solution, HQ, and they found that its pH was 3.7. Deduce, giving a reason, the strength (weak or strong) of the acid HQ.
Suggest a method, other than those mentioned above, that could be used to solve the problem and outline how the results would distinguish between a strong acid and a weak acid.
Markscheme

+ sign must be present. Do not award mark for incorrect notation 4, 4+, 3, 3+ etc.
Do not award M2 if inconsistent with M1.
increases / makes it stronger;
(more \({{\text{H}}^ + }\) would) drive/shift equilibrium to the right/towards products
(accepting more electrons);
\({\text{C}}{{\text{d}}^{2 + }}\);
Do not allow incorrect notation such as Cd, Cd(II), or Cd+2.
\({\text{2Ti(s)}} + {\text{3C}}{{\text{d}}^{2 + }}{\text{(aq)}} \to {\text{2T}}{{\text{i}}^{3 + }}{\text{(aq)}} + {\text{3Cd(s)}}\);
Ignore state symbols.
Allow ECF from (b)(i) for a correct equation.
\({\text{C}}{{\text{d}}^{2 + }}\);
Charge must be given.
Do not allow incorrect notation such as Cd, Cd(II), or Cd+2 but penalize
only once in b(i) and b(iii) .
Allow ECF, if Eu2+ is written both in part (i) and part (iii).
salt bridge;
Accept specific examples of salt bridges, such as filter paper dipped in aqueous KNO3.
allows the movement of ions (between the two solutions) / completes the circuit / maintains electrical neutrality;
Accept movement of charges/negative ions/positive ions.
donates \({{\text{H}}^ + }\)/protons;
strong acid completely/100%/fully dissociated/ionized and weak acid partially/slightly dissociated/ionized;
not a good choice / poor choice;
requires same volume of the base / the amount/volume to react/for neutralization does not depend on the acid strength;
sulfuric acid is diprotic/dibasic/liberates two protons/\({{\text{H}}^ + }\);
Accept “reacts with 2 moles of alkali/base”.
Strong acid: hydrochloric acid/HCl / nitric acid/\({\text{HN}}{{\text{O}}_{\text{3}}}\);
Weak acid: ethanoic acid/\({\text{C}}{{\text{H}}_{\text{3}}}{\text{COOH}}\);
Allow acetic acid for weak acid.
Accept any other strong/weak monobasic acids as appropriate.
Do not accept non-monobasic acids, such as phosphoric acid and carbonic acid.
weak;
strong \({\text{0.100 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) acid has a pH of 1/lower than that observed;
Accept “pH value of 3.7 means that it produces only 10–3.7/2.0 \( \times \) 10–4 [H+] in water”.
measure the rate of reaction with reactive metal/(metal) carbonate/metal oxide;
strong acid would react faster/more vigorously / weak acid would react slower/less vigorously;
Accept specific substances, such as Mg and CaCO3, which react with acids.
OR
measure conductivity;
higher for strong acid / lower for weak acid;
OR
measure heat/enthalpy of neutralization;
greater for strong acid / lower for weak acid;
Do not accept pH/universal indicator paper.
Examiners report
In part (a) (i), most candidates scored full marks although some candidates continue to write incorrect notation (4, 4+) for oxidation states.
In part (ii), some candidates missed the word equilibrium in the question and hence could not state that equilibrium will shift towards right and strength of oxidizing agent will increase.
In part (b) (i), (iii), the correct answer was \({\text{C}}{{\text{d}}^{2 + }}\) but many candidates wrote Cd, Eu or Ti.
In part (ii), the better candidates wrote the correct balanced chemical equation. Some included electrons in the equation which was surprising and some did not read the question where the reaction with Ti metal was asked.
In part (b) (i), (iii), the correct answer was \({\text{C}}{{\text{d}}^{2 + }}\) but many candidates wrote Cd, Eu or Ti.
In part (iv), many candidates identified the salt bridge but some missed the reference to the movement of ions.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
In part (c), most candidates were able to define a Bronsted-Lowry acid. The difference between strong and weak was usually correctly stated although only better candidates stated that strong acid is assumed to be 100% dissociated. Part (iii) proved to be difficult where very few candidates stated correctly that it is not a good choice because it requires the same volume of the base. Many candidates, however, knew the fact that sulfuric acid is diprotic in part (iv). In part (v), majority of candidates correctly identified the strong and weak acid whereas weaker candidates stated NaOH as a weak acid. Part (vi) was poorly done with many candidates stating pH 3.7 as strong acid. In part (vii), many candidates scored full marks but universal indicator paper was often suggested, which of course, scored no marks.
A hydrocarbon has the empirical formula \({{\text{C}}_{\text{3}}}{{\text{H}}_{\text{7}}}\). When 1.17 g of the compound is heated to 85 °C at a pressure of 101 kPa it occupies a volume of \({\text{400 c}}{{\text{m}}^{\text{3}}}\).
(i) Calculate the molar mass of the compound, showing your working.
(ii) Deduce the molecular formula of the compound.
\({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) exists as three isomers. Identify the structure of the isomer with the lowest boiling point and explain your choice.
Ethanol is a primary alcohol that can be oxidized by acidified potassium dichromate(VI). Distinguish between the reaction conditions needed to produce ethanal and ethanoic acid.
Ethanal:
Ethanoic acid:
Determine the oxidation number of carbon in ethanol and ethanal.
Ethanol:
Ethanal:
Deduce the half-equation for the oxidation of ethanol to ethanal.
Deduce the overall redox equation for the reaction of ethanol to ethanal with acidified potassium dichromate(VI) by combining your answer to part (c) (iii) with the following half-equation:
\[{\text{C}}{{\text{r}}_{\text{2}}}{\text{O}}_{\text{7}}^{2 - }{\text{(aq)}} + {\text{14}}{{\text{H}}^ + }{\text{(aq)}} + {\text{6}}{{\text{e}}^ - } \to {\text{2C}}{{\text{r}}^{3 + }}{\text{(aq)}} + {\text{7}}{{\text{H}}_{\text{2}}}{\text{O(l)}}\]
Describe two characteristics of a reaction at equilibrium.
Describe how a catalyst increases the rate of a reaction.
State and explain the effect of a catalyst on the position of equilibrium.
Ethanoic acid reacts with ethanol to form the ester ethyl ethanoate.
\[{\text{C}}{{\text{H}}_{\text{3}}}{\text{COOH(l)}} + {\text{C}}{{\text{H}}_{\text{3}}}{\text{C}}{{\text{H}}_{\text{2}}}{\text{OH(l)?????C}}{{\text{H}}_{\text{3}}}{\text{COOC}}{{\text{H}}_{\text{2}}}{\text{C}}{{\text{H}}_{\text{3}}}{\text{(l)}} + {{\text{H}}_{\text{2}}}{\text{O(l)}}\]
The esterification reaction is exothermic. State the effect of increasing temperature on the value of the equilibrium constant (\({K_{\text{c}}}\)) for this reaction.
Markscheme
(i) \({\text{temperature}} = 358{\text{ K}}\);
\(M = \frac{{mRT}}{{pV}}/1.17 \times 8.31 \times \frac{{358}}{{(0.40 \times 101)}}\);
\({\text{(}}M = {\text{) }}86.2{\text{ (gmo}}{{\text{l}}^{ - 1}}{\text{)}}\);
Award [1 max] for correct final answer without working.
(ii) \({{\text{C}}_6}{{\text{H}}_{14}}\);
\({\text{C(C}}{{\text{H}}_3}{{\text{)}}_4}\);
Accept correct name 2,2-dimethylpropane.
Do not penalize missing H atoms.
weakest London/dispersion/van der Waals’/vdW/instantaneous induced dipoleinduced dipole forces because of smallest surface area/contact
OR
weakest London/dispersion/van der Waals’/vdW/ instantaneous induced dipoleinduced dipole forces because of least distortion of the electron cloud
OR
weakest London/dispersion/van der Waals’/vdW/ instantaneous induced dipoleinduced dipole forces because polarizability of electrons (in electron cloud) is less;
Accept other words to that effect but student must mention a correct IMF and a correct reason.
Ethanal: distill off product as it forms;
Accept distillation.
Ethanoic acid: (heat under) reflux / use excess oxidizing agent;
Ethanol: –2/–II;
Ethanal: –1/–I;
Do not accept 2– or 1–, but penalize only once.
\({\text{C}}{{\text{H}}_3}{\text{C}}{{\text{H}}_2}{\text{OH}} \to {\text{C}}{{\text{H}}_3}{\text{CHO}} + {\text{2}}{{\text{H}}^ + } + {\text{2}}{{\text{e}}^ - }\);
Half-equation required. Do not accept \({C_2}{H_5}OH + 2[O] \to C{H_3}CHO + {H_2}O\).
Accept e for \({e^ - }\).
\({\text{3C}}{{\text{H}}_3}{\text{C}}{{\text{H}}_2}{\text{OH(aq)}} + {\text{C}}{{\text{r}}_2}{\text{O}}_7^{2 - }{\text{(aq)}} + {\text{8}}{{\text{H}}^ + }{\text{(aq)}} \to {\text{2C}}{{\text{r}}^{{\text{3}} + }}{\text{(aq)}} + {\text{3C}}{{\text{H}}_3}{\text{CHO(l)}} + {\text{7}}{{\text{H}}_2}{\text{O(l)}}\)
correct reactants and products;
correct balancing;
M2 can only be scored if M1 correct.
Ignore state symbols.
rate of forward process/reaction = rate of backward/reverse process/reaction;
concentrations of reactants and products remain constant;
no change in macroscopic properties;
closed/isolated system / constant matter/energy;
provides alternative pathway (of lower energy);
lowers activation energy (of the reaction) / more particles with \(E \geqslant {E_{\text{a}}}\);
no effect (on position of equilibrium);
increases rate of forward and reverse reactions (equally);
decreases;
Examiners report
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
This was the least popular question however many who chose it were successful in parts. Part (a) that required a calculation of \({M_{\text{r}}}\) was quite well done. However (b) that asked for the isomer of \({{\text{C}}_{\text{5}}}{{\text{H}}_{{\text{12}}}}\) with the lowest boiling point was not well answered. Identification of the methods to produce ethanal or ethanoic acid was done well by the strong candidates and others just guessed. Deduction of oxidation numbers and then writing of redox equations was not well answered. However (d) and (e) about equilibrium were answered well by many candidates, although there were again some very poor answers.
Urea, (H2N)2CO, is excreted by mammals and can be used as a fertilizer.
Calculate the percentage by mass of nitrogen in urea to two decimal places using section 6 of the data booklet.
Suggest how the percentage of nitrogen affects the cost of transport of fertilizers giving a reason.
The structural formula of urea is shown.

Predict the electron domain and molecular geometries at the nitrogen and carbon atoms, applying the VSEPR theory.

Urea can be made by reacting potassium cyanate, KNCO, with ammonium chloride, NH4Cl.
KNCO(aq) + NH4Cl(aq) → (H2N)2CO(aq) + KCl(aq)
Determine the maximum mass of urea that could be formed from 50.0 cm3 of 0.100 mol dm−3 potassium cyanate solution.
Urea can also be made by the direct combination of ammonia and carbon dioxide gases.
2NH3(g) + CO2(g) \( \rightleftharpoons \) (H2N)2CO(g) + H2O(g) ΔH < 0
Predict, with a reason, the effect on the equilibrium constant, Kc, when the temperature is increased.
Suggest one reason why urea is a solid and ammonia a gas at room temperature.
Sketch two different hydrogen bonding interactions between ammonia and water.
The combustion of urea produces water, carbon dioxide and nitrogen.
Formulate a balanced equation for the reaction.
The mass spectrum of urea is shown below.

Identify the species responsible for the peaks at m/z = 60 and 44.
The IR spectrum of urea is shown below.

Identify the bonds causing the absorptions at 3450 cm−1 and 1700 cm−1 using section 26 of the data booklet.
Predict the number of signals in the 1H NMR spectrum of urea.
Markscheme
molar mass of urea «= 4 × 1.01 + 2 × 14.01 + 12.01 + 16.00» = 60.07 «g mol–1»
«% nitrogen = \(\frac{{2 \times 14.01}}{{60.07}}\) × 100 =» 46.65 «%»
Award [2] for correct final answer.
Award [1 max] for final answer not to two decimal places.
[2 marks]
«cost» increases AND lower N% «means higher cost of transportation per unit of nitrogen»
OR
«cost» increases AND inefficient/too much/about half mass not nitrogen
Accept other reasonable explanations.
Do not accept answers referring to safety/explosions.
[1 mark]

Note: Urea’s structure is more complex than that predicted from VSEPR theory.
[3 marks]
n(KNCO) «= 0.0500 dm3 × 0.100 mol dm–3» = 5.00 × 10–3 «mol»
«mass of urea = 5.00 × 10–3 mol × 60.07 g mol–1» = 0.300 «g»
Award [2] for correct final answer.
[2 marks]
«Kc» decreases AND reaction is exothermic
OR
«Kc» decreases AND ΔH is negative
OR
«Kc» decreases AND reverse/endothermic reaction is favoured
[1 mark]
Any one of:
urea has greater molar mass
urea has greater electron density/greater London/dispersion
urea has more hydrogen bonding
urea is more polar/has greater dipole moment
Accept “urea has larger size/greater van der Waals forces”.
Do not accept “urea has greater intermolecular forces/IMF”.
[1 mark]

Award [1] for each correct interaction.
If lone pairs are shown on N or O, then the lone pair on N or one of the lone pairs on O MUST be involved in the H-bond.
Penalize solid line to represent H-bonding only once.
[2 marks]
2(H2N)2CO(s) + 3O2(g) → 4H2O(l) + 2CO2(g) + 2N2(g)
correct coefficients on LHS
correct coefficients on RHS
Accept (H2N)2CO(s) + \(\frac{3}{2}\)O2(g) → 2H2O(l) + CO2(g) + N2(g).
Accept any correct ratio.
[2 marks]
60: CON2H4+
44: CONH2+
Accept “molecular ion”.
[2 marks]
3450 cm–1: N–H
1700 cm–1: C=O
Do not accept “O–H” for 3450 cm–1.
[2 marks]
1
[1 mark]
Examiners report
Water is an important substance that is abundant on the Earth’s surface. Water dissociates according to the following equation.
\[{{\text{H}}_{\text{2}}}{\text{O(l)}} \rightleftharpoons {{\text{H}}^ + }{\text{(aq)}} + {\text{O}}{{\text{H}}^ - }{\text{(aq)}}\]
The graph below shows how the volume of carbon dioxide formed varies with time when a hydrochloric acid solution is added to excess calcium carbonate in a flask.

(i) State the equilibrium constant expression for the dissociation of water.
(ii) Explain why even a very acidic aqueous solution still has some \({\text{O}}{{\text{H}}^ - }\) ions present in it.
(iii) State and explain the effect of increasing temperature on the equilibrium constant above given that the dissociation of water is an endothermic process.
(iv) The pH of a solution is 2. If its pH is increased to 6, deduce how the hydrogen ion concentration changes.
In carbonated drinks containing dissolved carbon dioxide under high pressure, the
following dynamic equilibrium exists.
\[{\text{C}}{{\text{O}}_2}({\text{aq)}} \rightleftharpoons {\text{C}}{{\text{O}}_2}({\text{g)}}\]
Describe the effect of opening a carbonated drink container and outline how this
equilibrium is affected.
(i) Explain the shape of the curve.
(ii) Copy the above graph on your answer sheet and sketch the curve you would obtain if double the volume of hydrochloric acid solution of half the concentration as in the example above is used instead, with all other variables kept constant from the original. Explain why the shape of the curve is different.
(iii) Outline one other way in which the rate of this reaction can be studied in a school laboratory. Sketch a graph to illustrate how the selected variable would change with time.
(iv) Define the term activation energy and state one reason why the reaction between calcium carbonate and hydrochloric acid takes place at a reasonably fast rate at room temperature.
Markscheme
(i) \({K_{\text{c}}} = \frac{{{\text{[}}{{\text{H}}^ + }{\text{][O}}{{\text{H}}^ - }{\text{]}}}}{{{\text{[}}{{\text{H}}_2}{\text{O]}}}}/{K_{\text{c}}} = \frac{{{{{\text{[}}{{\text{H}}_3}{\text{O]}}}^ + }{\text{[O}}{{\text{H}}^ - }{\text{]}}}}{{{\text{[}}{{\text{H}}_2}{\text{O]}}}}/{K_{\text{w}}} = {\text{[}}{{\text{H}}^ + }{\text{][O}}{{\text{H}}^ - }{\text{]}}/{K_{\text{w}}} = {\text{[}}{{\text{H}}_{\text{3}}}{{\text{O}}^ + }{\text{][O}}{{\text{H}}^ - }{\text{]}}\);
Do not award mark if [ ] are omitted or other brackets are used.
Expression must be consistent with \({K_{\text{c}}}/{K_{\text{w}}}\).
(ii) \({\text{[}}{{\text{H}}^ + }{\text{]}}\) increases, \({\text{[O}}{{\text{H}}^ - }{\text{]}}\) decreases but still some present (\({K_{\text{w}}}/{K_{\text{c}}}\) constant) / \({\text{[O}}{{\text{H}}^ - }{\text{]}}\)
cannot go to zero as equilibrium present / \({\text{[O}}{{\text{H}}^ - }{\text{]}} = \frac{{{K_{\text{w}}}}}{{{\text{[}}{{\text{H}}^ + }{\text{]}}}}/\frac{{{K_{\text{c}}}{\text{[}}{{\text{H}}_2}{\text{O]}}}}{{{\text{[}}{{\text{H}}^ + }{\text{]}}}}\), thus \({\text{[O}}{{\text{H}}^ - }{\text{]}}\)
cannot be zero / OWTTE;
Accept equilibrium present.
(iii) (changing T disturbs equilibrium) forward reaction favoured / equilibrium shifts to the right;
to use up (some of the) heat supplied;
(\({K_{\text{w}}}/{K_{\text{c}}}\)) increases (as both \({\text{[}}{{\text{H}}^ + }{\text{]}}\) and \({\text{[O}}{{\text{H}}^ - }{\text{]}}\) increase);
(iv) \({\text{pH}} = 2{\text{, [}}{{\text{H}}^ + }{\text{]}} = 0.01{\text{ mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) and \({\text{pH}} = 6{\text{, [}}{{\text{H}}^ + }{\text{]}} = {10^{ - 6}}{\text{ mol}}\,{\text{d}}{{\text{m}}^{ - 3}}/{\text{[}}{{\text{H}}^ + }{\text{]}} = {10^{ - {\text{pH}}}}\);
\({\text{[}}{{\text{H}}^ + }{\text{]}}\) decreased/changed by \({\text{10000/1}}{{\text{0}}^{ - 4}}\);
Award [2] for correct final answer.
\({\text{C}}{{\text{O}}_2}({\text{g)}}\) /gas escapes / (gas) pressure / \({\text{C}}{{\text{O}}_2}\) (above liquid) decreases / bubbles (of \({\text{C}}{{\text{O}}_2}\) gas) form in the liquid;
equilibrium shifts to the right (to replace the lost \({\text{C}}{{\text{O}}_2}\) gas);
(i) rate = increase in \(\frac{{{\text{volume}}}}{{{\text{time}}}}\) = slope of graph;
initially/to begin with steeper slope / fastest rate / volume of gas/ \({\text{C}}{{\text{O}}_2}\) produced faster/quickly as concentration of HCl highest / OWTTE;
as reaction progresses/with time, less steep slope / volume of gas production slows / rate decreases due to less frequent collisions as concentration (of HCl) decreases / OWTTE;
curve flattens/becomes horizontal when HCl used up/consumed (as there are no more \({{\text{H}}^ + }\) ions to collide with the \({\text{CaC}}{{\text{O}}_3}\) particles);
Each mark requires explanation.
(ii) 
less steep curve;
same maximum volume at later time;
half/lower \({{\text{H}}^ + }\)/acid concentration less frequent collisions slower rate;
same amount of HCl, same volume \({\text{C}}{{\text{O}}_2}\) produced;
(iii) mass loss/of \({\text{C}}{{\text{O}}_2}\) / mass of flask + content;
 ;
;
OR
 ;
;
OR
 ;
;
Do not penalize for missing x-axis label or for missing units on y-axis.
Accept if line meets time axis.
Award [1 max] if temperature is on the vertical axis and magnitude of slope decreases with time.
(iv) minimum/least energy (of colliding particles) for a reaction to occur / OWTTE;
low/lower \({E_{\text{a}}}\) /activation energy / greater/larger surface area/contact between \({\text{CaC}}{{\text{O}}_{\text{3}}}\) and HCl / high/higher HCl concentration/[HCl] / (sufficient) particles/molecules have activation energy;
Examiners report
This was the most popular question in Section B but responses were mixed. Part (a) was generally well dealt with but some candidates confused \({K_{\text{w}}}\) with \({K_{\text{c}}}\) or forgot to include charges on the ions in the equilibrium constant expression. Few received the mark for question (ii) although some mentioned equilibrium which was sufficient.
Candidates recognised that increasing the temperature shifts the equilibrium to the right, but most did not explain why, namely to use up some of the heat supplied. The calculation in (iv) was quite well done although some only gave a qualitative answer.
The equilibrium of carbonated drinks was well understood.
In part (c) (i) candidates frequently described the shape of the curve instead of offering an explanation using collisions theory. Candidates did state, for example, that the curve flattens but did not refer to consumption of HCl(aq), the limiting reagent. Only the better candidates were only able to link slope with rate and some still consider the rate to increase after the reaction has started. In (ii) most realised that the curve would be less steep but few drew a curve with the same maximum volume produced at a later time. Even fewer candidates were able to explain why the number of moles of carbon dioxide remained the same. Although some candidates chose mass loss / pH / pressure as the dependant variable in c(iii), some were penalised for imprecise answers such as mass of reactants without referring to mass of flask. Others misunderstood the question and described experiments that they had done with catalysis or described changes with temperature as the dependant variable. (c)(iv) was generally well answered, but again some responses lacked precision; the activation energy is the minimum energy needed for a reaction to occur.
A mixture of 1.00 mol SO2(g), 2.00 mol O2(g) and 1.00 mol SO3(g) is placed in a 1.00 dm3 container and allowed to reach equilibrium.
2SO2(g) + O2(g) \( \rightleftharpoons \) 2SO3(g)
Distinguish between the terms reaction quotient, Q, and equilibrium constant, Kc.
The equilibrium constant, Kc, is 0.282 at temperature T.
Deduce, showing your work, the direction of the initial reaction.
Markscheme
Q: non-equilibrium concentrations AND Kc: equilibrium concentrations
OR
Q: «measured» at any time AND Kc: «measured» at equilibrium
[1 mark]
«Q = \(\frac{{{{[{\text{S}}{{\text{O}}_3}]}^2}}}{{{{[{\text{S}}{{\text{O}}_2}]}^2}[{{\text{O}}_2}]}} = \frac{{{{1.00}^2}}}{{{{1.00}^2} \times 2.00}}\)» = 0.500
reverse reaction favoured/reaction proceeds to the left AND Q > Kc/0.500 > 0.282
Do not award M2 without M1.
[2 marks]
Examiners report
Chemical equilibrium and kinetics are important concepts in chemistry.
The oxidation of sulfur dioxide is an important reaction in the Contact process used to manufacture sulfuric acid.
\[\begin{array}{*{20}{l}} {{\text{2S}}{{\text{O}}_2}{\text{(g)}} + {{\text{O}}_{\text{2}}}{\text{(g)}} \rightleftharpoons {\text{2S}}{{\text{O}}_{\text{3}}}{\text{(g)}}}&{\Delta H = - 198.2{\text{ kJ}}} \end{array}\]
Vanadium(V) oxide, \({{\text{V}}_{\text{2}}}{{\text{O}}_{\text{5}}}\), is a catalyst that can be used in the Contact process. It provides an alternative pathway for the reaction, lowering the activation energy, \({E_{\text{a}}}\).
A glass container is half-filled with liquid bromine and then sealed. The system eventually reaches a dynamic equilibrium. State one characteristic of a system in equilibrium.
(i) Deduce the equilibrium constant expression, \({K_{\text{c}}}\).
(ii) Predict how each of the following changes affects the position of equilibrium and
the value of \({K_{\text{c}}}\).

(i) Define the term activation energy, \({E_{\text{a}}}\).
(ii) Sketch the two Maxwell–Boltzmann energy distribution curves for a fixed amount of gas at two different temperatures, \({T_1}\) and \({T_2}{\text{ }}({T_2} > {T_1})\). Label both axes.

Markscheme
rate of forward process/reaction = rate of backwards/reverse process/reaction / rate of vaporization/evaporation = rate of condensation;
concentrations of reactants and products remain constant;
no change in macroscopic properties / closed system / constant matter/energy / OWTTE;
Do not accept concentration of reactants and products are equal.
Accept constant colour of \({\rm{B}}{{\rm{r}}_2}\) vapour/liquid.
(i) \(({K_{\text{c}}} = )\frac{{{{{\text{[S}}{{\text{O}}_3}{\text{]}}}^2}}}{{{{{\text{[S}}{{\text{O}}_2}{\text{]}}}^2}{\text{[}}{{\text{O}}_2}{\text{]}}}}\);
(ii) 
Award [1] for any two or three correct, [2] for any four or five correct, [3] for six correct.
(i) minimum/least/smallest energy needed (by reactants/colliding particles) to react/start/initiate a reaction;
Allow energy difference between reactants and transition state.
(ii) x-axis label: (kinetic) energy/(K)E and y-axis label: probability/fraction of molecules/particles / probability density;
Allow number of molecules/particles for y-axis.
correct shape of a typical Maxwell–Boltzmann energy distribution curve;
Do not award mark if curve is symmetric, does not start at zero or if it crosses x-axis.
two curves represented with second curve for \({T_2} > {T_1}\) to right of first curve, peak maximum lower than first curve and after the curves cross going to the right, \({T_2}\) curve needs to be above \({T_1}\) curve as illustrated;
M2 and M3 can be scored independently.

Examiners report
The equilibrium question was generally well answered, but some candidates suggested that the forward reaction equal the reverse reaction without reference to the rates, while some other candidates incorrectly stated that the concentration of reactants and products are equal.
In part (b) (i), the \({K_{\text{c}}}\) expression was usually written correctly. Part (b) (ii) was done well and candidates showed a good understanding of the effect of temperature and catalyst on an equilibrium system; however, weaker candidates incorrectly identified a change in the value of \({K_{\text{c}}}\) on increasing the pressure.
In part (c), the word minimum was often missed in the definition of activation energy. In the Maxwell–Boltzmann energy distribution curves, many candidates labelled the axes incorrectly. Also in some cases, the curves did not start at the origin or the curve for \({T_2}\) was drawn incorrectly at the same level on the y-axis. The weaker students drew an enthalpy level diagram instead of a Maxwell- Boltzmann distribution.
PCl5(g) and Cl2(g) were placed in a sealed flask and allowed to reach equilibrium at 200 °C. The enthalpy change, ΔH, for the decomposition of PCl5(g) is positive.

Deduce the equilibrium constant expression, Kc, for the decomposition of PCl5(g).
Deduce, giving a reason, the factor responsible for establishing the new equilibrium after 14 minutes.
Deduce the Lewis (electron dot) structure and molecular geometry of PCl3.
Markscheme
«Kc» = \(\frac{{\left[ {{\text{PC}}{{\text{l}}_3}} \right]\left[ {{\text{C}}{{\text{l}}_2}} \right]}}{{\left[ {{\text{PC}}{{\text{l}}_5}} \right]}}\)
[1 mark]
decrease in temperature
endothermic «reaction» AND «equilibrium» shifts to the left/reactants
OR
endothermic «reaction» AND Kc decreases
OR
endothermic «reaction» AND concentration of PCl5 increased/concentration of PCl3 and Cl2 decreased
OR
«equilibrium» shifts in exothermic direction
Do not accept “temperature change”.
Accept “ΔH positive” in place of “endothermic”.
Accept “products” instead of “PCl3 and Cl2”.
[2 marks]
Lewis structure:
Molecular geometry:
trigonal/triangular pyramidal
Penalize missing lone pairs once only between this question and 4(b).
Accept any combination of lines, dots or crosses to represent electrons.
Do not apply ECF.
[2 marks]
Examiners report
Two hydrides of nitrogen are ammonia and hydrazine, N2H4. One derivative of ammonia is methanamine whose molecular structure is shown below.
Hydrazine is used to remove oxygen from water used to generate steam or hot water.
N2H4(aq) + O2(aq) → N2(g) + 2H2O(l)
The concentration of dissolved oxygen in a sample of water is 8.0 × 10−3 g\(\,\)dm−3.
Estimate the H−N−H bond angle in methanamine using VSEPR theory.
Ammonia reacts reversibly with water.
NH3(g) + H2O(l) \( \rightleftharpoons \) NH4+(aq) + OH−(aq)
Explain the effect of adding H+(aq) ions on the position of the equilibrium.
Hydrazine reacts with water in a similar way to ammonia. Deduce an equation for the reaction of hydrazine with water.
Outline, using an ionic equation, what is observed when magnesium powder is added to a solution of ammonium chloride.
Hydrazine has been used as a rocket fuel. The propulsion reaction occurs in several stages but the overall reaction is:
N2H4(l) → N2(g) + 2H2(g)
Suggest why this fuel is suitable for use at high altitudes.
Determine the enthalpy change of reaction, ΔH, in kJ, when 1.00 mol of gaseous hydrazine decomposes to its elements. Use bond enthalpy values in section 11 of the data booklet.
N2H4(g) → N2(g) + 2H2(g)
The standard enthalpy of formation of N2H4(l) is +50.6 kJ\(\,\)mol−1. Calculate the enthalpy of vaporization, ΔHvap, of hydrazine in kJ\(\,\)mol−1.
N2H4(l) → N2H4(g)
(If you did not get an answer to (f), use −85 kJ but this is not the correct answer.)
Calculate, showing your working, the mass of hydrazine needed to remove all the dissolved oxygen from 1000 dm3 of the sample.
Calculate the volume, in dm3, of nitrogen formed under SATP conditions. (The volume of 1 mol of gas = 24.8 dm3 at SATP.)
Markscheme
107°
Accept 100° to < 109.5°.
Literature value = 105.8°
[1 mark]
removes/reacts with OH−
moves to the right/products «to replace OH− ions»
Accept ionic equation for M1.
[2 marks]
N2H4(aq) + H2O(l) \( \rightleftharpoons \) N2H5+(aq) + OH–(aq)
Accept N2H4(aq) + 2H2O(l) \( \rightleftharpoons \) N2H62+(aq) + 2OH–(aq).
Equilibrium sign must be present.
[1 mark]
bubbles
OR
gas
OR
magnesium disappears
2NH4+(aq) + Mg(s) → Mg2+(aq) + 2NH3(aq) + H2(g)
Do not accept “hydrogen” without reference to observed changes.
Accept "smell of ammonia".
Accept 2H+(aq) + Mg(s) → Mg2+(aq) + H2(g)
Equation must be ionic.
[2 mark]
no oxygen required
[1 mark]
bonds broken:
E(N–N) + 4E(N–H)
OR
158 «kJ\(\,\)mol–1» + 4 x 391 «kJ\(\,\)mol–1» / 1722 «kJ»
bonds formed:
E(N≡N) + 2E(H–H)
OR
945 «kJ\(\,\)mol–1» + 2 x 436 «kJ\(\,\)mol–1» / 1817 «kJ»
«ΔH = bonds broken – bonds formed = 1722 – 1817 =» –95 «kJ»
Award [3] for correct final answer.
Award [2 max] for +95 «kJ».
[3 marks]
OR
ΔHvap= −50.6 kJ\(\,\)mol−1 − (−95 kJ\(\,\)mol−1)
«ΔHvap =» +44 «kJ\(\,\)mol−1»
Award [2] for correct final answer.
Award [1 max] for −44 «kJ\(\,\)mol−1».
Award [2] for:
ΔHvap − = 50.6 kJ\(\,\)mol−1 − (−85 kJ\(\,\)mol−1) + = 34 «kJ\(\,\)mol−1».
Award [1 max] for −34 «kJ\(\,\)mol−1».
[2 marks]
total mass of oxygen «= 8.0 x 10–3 g\(\,\)dm–3 x 1000 dm3» = 8.0 «g»
n(O2) «\( = \frac{{8.0{\text{ g}}}}{{32.00{\text{ g}}\,{\text{mo}}{{\text{l}}^{ - 1}}}} = \)» 0.25 «mol»
OR
n(N2H4) = n(O2)
«mass of hydrazine = 0.25 mol x 32.06 g\(\,\)mol–1 =» 8.0 «g»
Award [3] for correct final answer.
[3 marks]
«n(N2H4) = n(O2) \( = \frac{{8.0{\text{ g}}}}{{32.00{\text{ g}}\,{\text{mo}}{{\text{l}}^{ - 1}}}} = \)» 0.25 «mol»
«volume of nitrogen = 0.25 mol x 24.8 dm3\(\,\)mol–1» = 6.2 «dm3»
Award [1] for correct final answer.
[1 mark]
Examiners report
Many reactions are in a state of equilibrium.
The equations for two acid-base reactions are given below.
HCO3– (aq) + H2O (l) \( \rightleftharpoons \) H2CO3 (aq) + OH– (aq)
HCO3– (aq) + H2O (l) \( \rightleftharpoons \) CO32– (aq) + H3O+ (aq)
The following reaction was allowed to reach equilibrium at 761 K.
H2 (g) + I2 (g) \( \rightleftharpoons \) 2HI (g) ΔHθ < 0
Outline the effect, if any, of each of the following changes on the position of equilibrium, giving a reason in each case.
Identify two different amphiprotic species in the above reactions.
State what is meant by the term conjugate base.
State the conjugate base of the hydroxide ion, OH–.
A student working in the laboratory classified HNO3, H2SO4, H3PO4 and HClO4 as acids based on their pH. He hypothesized that “all acids contain oxygen and hydrogen”.
Evaluate his hypothesis.
Markscheme
Award [1 max] if both effects are correct.
Reason for increasing volume:
Accept “concentration of all reagents reduced by an equal amount so cancels out in Kc expression”.
Accept “affects both forward and backward rates equally”.
HCO3– AND H2O
species that has one less proton/H+ ion «than its conjugate acid»
OR
species that forms its conjugate acid by accepting a proton
OR
species that is formed when an acid donates a proton
Do not accept “differs by one proton/H+ from conjugate acid”.
oxide ion/O2–
insufficient data to make generalization
OR
need to consider a «much» larger number of acids
OR
hypothesis will continue to be tested with new acids to see if it can stand the test of time
«hypothesis is false as» other acids/HCl/HBr/HCN/transition metal ion/BF3 do not contain oxygen
OR
other acids/HCl/HBr/HCN/transition metal ion/BF3 falsify hypothesis
correct inductive reasoning «based on limited sample»
«hypothesis not valid as» it contradicts current/accepted theories/Brønsted-Lowry/Lewis theory
[Max 2 Marks]
Examiners report
Phosgene, COCl2, is usually produced by the reaction between carbon monoxide and chlorine according to the equation:
(i) Deduce the equilibrium constant expression, Kc, for this reaction.
(ii) State the effect of an increase in the total pressure on the equilibrium constant, Kc.
(i) Sketch the potential energy profile for the synthesis of phosgene, using the axes given, indicating both the enthalpy of reaction and activation energy.
(ii) This reaction is normally carried out using a catalyst. Draw a dotted line labelled “Catalysed” on the diagram above to indicate the effect of the catalyst.
(iii) Sketch and label a second Maxwell–Boltzmann energy distribution curve representing the same system but at a higher temperature, Thigher.
(iv) Explain why an increase in temperature increases the rate of this reaction.
Markscheme
(i)
\( \ll {K_{\rm{c}}} = \gg \frac{{\left[ {{\rm{COC}}{{\rm{l}}_{\rm{2}}}} \right]}}{{\left[ {{\rm{CO}}} \right]\left[ {{\rm{C}}{{\rm{l}}_2}} \right]}}\)
(ii)
no effect
(i)
products lower than reactants AND enthalpy of reaction correctly marked and labelled with name or value
activation energy correctly marked and labelled with name or value
Accept other clear ways of indicating energy/ enthalpy changes.
(ii)
lower dotted curve, between same reactants and product levels, labelled “Catalysed”
(iii)
second curve at a higher temperature is correctly drawn (maximum lower and to right of original)
(iv)
greater proportion of molecules have E ≥ Ea or E >Ea
OR
greater area under curve to the right of the Ea
greater frequency of collisions «between molecules»
OR
more collisions per unit time/second
Do not accept just particles have greater kinetic energy.
Do not accept just “more collisions”.